微粒子合成化学・講義
|
|
|
- ぎんと つちかね
- 5 years ago
- Views:
Transcription
1 1
2 2
3 1 mol/l KCl 3
4 4
5 Derjaguin Landau Verway Overbeek B.V.Derjaguin and L.Landau;Acta Physicochim.,URSS, 14, E.J.W.Verwey and J.Th G Overbeek; Theory of the Stability of Lyophobic Colloids,
6 6
7 7
8 8
9 9
10 10
11 11
12 12
13 13
14 14
15 -Si-O-H -Si-O + H + 15
16 16
17 17
18 18
19 19
20 0 Helmholtz 0 20
21 0 Gouy-Chapman 0 21
22 0 Stern Stern Stern 0 Slip 22
23 23
24 0=Stern d 0 24
25 25
26 z+ eψ n+ = n + exp (1) 0 kt z eψ n = n0 exp kt n: n 0 : z: k: T: ψ: +,-: 26
27 ψ0 c RT c ψ 0 = ln zf c (2) R: c 0 : c at ψ 0 =
28 Poisson ψ ψ ψ ρ Δ ψ = div (grad ψ ) = + + = x y z ε ε ε r : ε 0 : ρ: r 0 (3) 28
29 29 ρ: n n n z z z = = = = , = = = + kt ze nze kt ze kt ze nze n n ze ψ ψ ψ ρ sinh 2 exp exp ) ( (4)
30 Poisson-Boltzmann (3),(4) x 2 d ψ 2nze zeψ = sinh (5) 2 dx ε ε kt r 0 (5) zeψ zeψ 0 tanh = tanh exp( κx) 4kT 4kT (6) 30
31 zeψ kt <<1 (5) d dx 2 ψ 2 2 = κ ψ κ = nz e ε ε kt r κ = z c (7) ψ = ψ exp( κ ) 0 x (7) (8) (9) (10) Debye-Huckel 31
32 32
33 : h P P = P E + P O P P E O = = ( n ε + r ε n 2 dψ dx ) kt 2nkT (15) (16) 33
34 P O P E ψ 0 P E (1) (16) P O P R (h) P R zeψ h / 2 ( h) = 2nkT cosh 1 kt ψ 2/h : (17) 34
35 ψ h/2 ψ s(h/2) zeψ / 4kT << 1 then tanh( zeψ / 4kT) zeψ / 4kT (6) ψ<20 mv ψ γ ( h / 2) = 8kT h = γ exp κ ze 2 zeψ 0 tanh 4kT (18) (19) 35
36 (17) ze ψ 2 h / 2 / kt << 1 then PR ( h) nkt{ zeψ h / 2 / kt} (18) κh>1 h cosh y 1 + y 2 P R 2 ( h) = 64nkTγ exp( κh) (20) 36
37 V R ( h) h = P R ( h) dh = 64nkT κ γ 2 exp( κ h) (21) 37
38 38
39 Derjaguin Derjaguin : a 1 a 2 H H<<a 1,a 2 P R ( H ) = 2π a a 1 2 V ( a H 1 a R + 2 (21) (22) a 1 =a 2 =a P R ( H ) = 64 π ankt κ γ 2 exp( ) κ h) (22) (23) 39
40 a V R ( H H ) = PR ( H ) dh 64πankT 2 = γ exp( κh) (24) 2 κ 40
41 zeψ 0 / 4kT << 1 then tanh( zeψ 0 / 4kT) zeψ 0 / 4kT (23),(24) zeψ 0 =4kT 1:1 25 ψ 0 =103 mv ψ 0 =20 mv zeψ 0 /4kT tanh{ zeψ 0 /4kT} 1% P V R R 20mV ( H ( H ) ) = = 2 2 π π a a ε ε r r ε ε (13) 2 0κψ 0 exp( κh) 2 0ψ 0 exp( κh) (25) (26) 41
42 42 ) exp( 2 ) ( h a H P r R κ κψ ε ε π = ) exp( 2 ) ( h a H V r R κ ψ ε ε π = (13) ) exp( 2 ) ( 0 2 H a H P r R κ ε κε σ π = ) exp( 2 ) ( H a H V r R κ ε ε κ σ π = (25) (26) (27) (28) κψ ε ε σ r = (13)
43 van der Waals aa P A ( H ) = 12H aa V A ( H ) = 12H A Hamaker 2 (29) (30) 43
44 ) exp( 2 ) ( H aa H a H P r T = κ ε κε σ π H aa H a H V r T 12 ) exp( 2 ) ( = κ ε ε κ σ π H aa h a H V r T 12 ) exp( 2 ) ( = κ ψ ε ε π (31) (32) (33)
45 45
46 V T ( H ) = 2 π a ε r ε ψ 2 0 κ 0 exp( H ) aa 12 H ε r, ε 0, ψ 0, A a κ 46
47 2 2 κ = ε nz r ε 0 2 e 2 kt e ε r ε 0 k n z T 47
48 n z T κ 48
49 V T ( H ) 2 a r 0 0 exp( H ) = π ε ε ψ κ 2 aa 12 H 49
50 van der Waals 50
51 51
52 van der Waals 52
53 53
54 54
55 55
56 56
57 57
58 58
59 59
60 60
61 61
62 62
63 63
64 64
65 65
66 66
67 67
68 68
69 KCl 1 mol/l KCl 69
70 70
71 71
72 72
73 73
74 74
75 75
76 76
77 77
78 78
79 79
80 80
81 81
82 82
83 83
84 84
85 85
86 86
87 87
88 88
89 89
90 90
91 91
92 92
93 93
94 94
95 95
96 2,3,7,8- ppm O- n- cm/day ppb 2 96
97 97
98 98
99 99
100 No data
101 (8.01%) 49 (16.67%) 242 (3.62%) 59 (0.81%) 1 (0.34%) 2 (0.03%) 54 (0.74%) 11 (3.74%) 26 (0.39%) 914 (12.47%) (4.65%) 81 (1.11%) 16 (5.44%) 29 (0.43%) 1614 (22.03%) 61 (20.75%) 581 (8.68%)
102 (A) [%] (B) [%] B/A
103 103
104 104
105 105
106 gteq/ ( ) ( )
107 107
108 108
109 109
110 ( C) (ng-teq/nm 3 ) ( 1111)
111 111
112 112
113 113
114 114
115 115
微粒子合成化学・講義
 http://www.tagen.tohoku.ac.jp/labo/muramatsu/mura/main.html E-mail: mura@tagen.tohoku.ac.jp 1 Derjaguin Landau Verway Overbeek B.V.Derjaguin and L.Landau;Acta Physicochim.,URSS, 14, 633 1941. E.J.W.Verwey
http://www.tagen.tohoku.ac.jp/labo/muramatsu/mura/main.html E-mail: mura@tagen.tohoku.ac.jp 1 Derjaguin Landau Verway Overbeek B.V.Derjaguin and L.Landau;Acta Physicochim.,URSS, 14, 633 1941. E.J.W.Verwey
コロイド化学と界面化学
 環境表面科学講義 http://res.tagen.tohoku.ac.jp/~liquid/mura/kogi/kaimen/ E-mail: mura@tagen.tohoku.ac.jp 村松淳司 分散と凝集 ( 平衡論的考察! 凝集! van der Waals 力による相互作用! 分散! 静電的反発力 凝集 分散! 粒子表面の電位による反発 分散と凝集 考え方! van der Waals
環境表面科学講義 http://res.tagen.tohoku.ac.jp/~liquid/mura/kogi/kaimen/ E-mail: mura@tagen.tohoku.ac.jp 村松淳司 分散と凝集 ( 平衡論的考察! 凝集! van der Waals 力による相互作用! 分散! 静電的反発力 凝集 分散! 粒子表面の電位による反発 分散と凝集 考え方! van der Waals
ナノ粒子のサイズ・形態制御と 構造敏感型触媒プロセスへの応用
 環境表面科学講義 http://res.tagen.tohoku.ac.jp/~liquid/mura/kogi/kaimen/ E-mail: mura@tagen.tohoku.ac.jp 村松淳司 DLVO 理論 分散と凝集をどう扱うか それぞれを別の 2 つの力とする 3 分散と凝集 考え方 V total = V H + V el V H : van der Waals 力による相互作用エネルギー
環境表面科学講義 http://res.tagen.tohoku.ac.jp/~liquid/mura/kogi/kaimen/ E-mail: mura@tagen.tohoku.ac.jp 村松淳司 DLVO 理論 分散と凝集をどう扱うか それぞれを別の 2 つの力とする 3 分散と凝集 考え方 V total = V H + V el V H : van der Waals 力による相互作用エネルギー
. ev=,604k m 3 Debye ɛ 0 kt e λ D = n e n e Ze 4 ln Λ ν ei = 5.6π / ɛ 0 m/ e kt e /3 ν ei v e H + +e H ev Saha x x = 3/ πme kt g i g e n
 003...............................3 Debye................. 3.4................ 3 3 3 3. Larmor Cyclotron... 3 3................ 4 3.3.......... 4 3.3............ 4 3.3...... 4 3.3.3............ 5 3.4.........
003...............................3 Debye................. 3.4................ 3 3 3 3. Larmor Cyclotron... 3 3................ 4 3.3.......... 4 3.3............ 4 3.3...... 4 3.3.3............ 5 3.4.........
I-2 (100 ) (1) y(x) y dy dx y d2 y dx 2 (a) y + 2y 3y = 9e 2x (b) x 2 y 6y = 5x 4 (2) Bernoulli B n (n = 0, 1, 2,...) x e x 1 = n=0 B 0 B 1 B 2 (3) co
 16 I ( ) (1) I-1 I-2 I-3 (2) I-1 ( ) (100 ) 2l x x = 0 y t y(x, t) y(±l, t) = 0 m T g y(x, t) l y(x, t) c = 2 y(x, t) c 2 2 y(x, t) = g (A) t 2 x 2 T/m (1) y 0 (x) y 0 (x) = g c 2 (l2 x 2 ) (B) (2) (1)
16 I ( ) (1) I-1 I-2 I-3 (2) I-1 ( ) (100 ) 2l x x = 0 y t y(x, t) y(±l, t) = 0 m T g y(x, t) l y(x, t) c = 2 y(x, t) c 2 2 y(x, t) = g (A) t 2 x 2 T/m (1) y 0 (x) y 0 (x) = g c 2 (l2 x 2 ) (B) (2) (1)
master.dvi
 4 Maxwell- Boltzmann N 1 4.1 T R R 5 R (Heat Reservor) S E R 20 E 4.2 E E R E t = E + E R E R Ω R (E R ) S R (E R ) Ω R (E R ) = exp[s R (E R )/k] E, E E, E E t E E t E exps R (E t E) exp S R (E t E )
4 Maxwell- Boltzmann N 1 4.1 T R R 5 R (Heat Reservor) S E R 20 E 4.2 E E R E t = E + E R E R Ω R (E R ) S R (E R ) Ω R (E R ) = exp[s R (E R )/k] E, E E, E E t E E t E exps R (E t E) exp S R (E t E )
( )
 7..-8..8.......................................................................... 4.................................... 3...................................... 3..3.................................. 4.3....................................
7..-8..8.......................................................................... 4.................................... 3...................................... 3..3.................................. 4.3....................................
, 1.,,,.,., (Lin, 1955).,.,.,.,. f, 2,. main.tex 2011/08/13( )
 81 4 2 4.1, 1.,,,.,., (Lin, 1955).,.,.,.,. f, 2,. 82 4.2. ζ t + V (ζ + βy) = 0 (4.2.1), V = 0 (4.2.2). (4.2.1), (3.3.66) R 1 Φ / Z, Γ., F 1 ( 3.2 ). 7,., ( )., (4.2.1) 500 hpa., 500 hpa (4.2.1) 1949,.,
81 4 2 4.1, 1.,,,.,., (Lin, 1955).,.,.,.,. f, 2,. 82 4.2. ζ t + V (ζ + βy) = 0 (4.2.1), V = 0 (4.2.2). (4.2.1), (3.3.66) R 1 Φ / Z, Γ., F 1 ( 3.2 ). 7,., ( )., (4.2.1) 500 hpa., 500 hpa (4.2.1) 1949,.,
4.6 (E i = ε, ε + ) T Z F Z = e βε + e β(ε+ ) = e βε (1 + e β ) F = kt log Z = kt log[e βε (1 + e β )] = ε kt ln(1 + e β ) (4.18) F (T ) S = T = k = k
![4.6 (E i = ε, ε + ) T Z F Z = e βε + e β(ε+ ) = e βε (1 + e β ) F = kt log Z = kt log[e βε (1 + e β )] = ε kt ln(1 + e β ) (4.18) F (T ) S = T = k = k 4.6 (E i = ε, ε + ) T Z F Z = e βε + e β(ε+ ) = e βε (1 + e β ) F = kt log Z = kt log[e βε (1 + e β )] = ε kt ln(1 + e β ) (4.18) F (T ) S = T = k = k](/thumbs/94/119235351.jpg) 4.6 (E i = ε, ε + ) T Z F Z = e ε + e (ε+ ) = e ε ( + e ) F = kt log Z = kt loge ε ( + e ) = ε kt ln( + e ) (4.8) F (T ) S = T = k = k ln( + e ) + kt e + e kt 2 + e ln( + e ) + kt (4.20) /kt T 0 = /k (4.20)
4.6 (E i = ε, ε + ) T Z F Z = e ε + e (ε+ ) = e ε ( + e ) F = kt log Z = kt loge ε ( + e ) = ε kt ln( + e ) (4.8) F (T ) S = T = k = k ln( + e ) + kt e + e kt 2 + e ln( + e ) + kt (4.20) /kt T 0 = /k (4.20)
H22環境地球化学4_化学平衡III_ ppt
 1 2 3 2009年度 環境地球化学 大河内 温度上昇による炭酸水の発泡 気泡 温度が高くなると 溶けきれなくなった 二酸化炭素が気泡として出てくる 4 2009年度 環境地球化学 圧力上昇による炭酸水の発泡 栓を開けると 瓶の中の圧力が急激に 小さくなるので 発泡する 大河内 5 CO 2 K H CO 2 H 2 O K H + 1 HCO 3- K 2 H + CO 3 2- (M) [CO
1 2 3 2009年度 環境地球化学 大河内 温度上昇による炭酸水の発泡 気泡 温度が高くなると 溶けきれなくなった 二酸化炭素が気泡として出てくる 4 2009年度 環境地球化学 圧力上昇による炭酸水の発泡 栓を開けると 瓶の中の圧力が急激に 小さくなるので 発泡する 大河内 5 CO 2 K H CO 2 H 2 O K H + 1 HCO 3- K 2 H + CO 3 2- (M) [CO
No δs δs = r + δr r = δr (3) δs δs = r r = δr + u(r + δr, t) u(r, t) (4) δr = (δx, δy, δz) u i (r + δr, t) u i (r, t) = u i x j δx j (5) δs 2
 No.2 1 2 2 δs δs = r + δr r = δr (3) δs δs = r r = δr + u(r + δr, t) u(r, t) (4) δr = (δx, δy, δz) u i (r + δr, t) u i (r, t) = u i δx j (5) δs 2 = δx i δx i + 2 u i δx i δx j = δs 2 + 2s ij δx i δx j
No.2 1 2 2 δs δs = r + δr r = δr (3) δs δs = r r = δr + u(r + δr, t) u(r, t) (4) δr = (δx, δy, δz) u i (r + δr, t) u i (r, t) = u i δx j (5) δs 2 = δx i δx i + 2 u i δx i δx j = δs 2 + 2s ij δx i δx j
30
 3 ............................................2 2...........................................2....................................2.2...................................2.3..............................
3 ............................................2 2...........................................2....................................2.2...................................2.3..............................
( ) ,
 II 2007 4 0. 0 1 0 2 ( ) 0 3 1 2 3 4, - 5 6 7 1 1 1 1 1) 2) 3) 4) ( ) () H 2.79 10 10 He 2.72 10 9 C 1.01 10 7 N 3.13 10 6 O 2.38 10 7 Ne 3.44 10 6 Mg 1.076 10 6 Si 1 10 6 S 5.15 10 5 Ar 1.01 10 5 Fe 9.00
II 2007 4 0. 0 1 0 2 ( ) 0 3 1 2 3 4, - 5 6 7 1 1 1 1 1) 2) 3) 4) ( ) () H 2.79 10 10 He 2.72 10 9 C 1.01 10 7 N 3.13 10 6 O 2.38 10 7 Ne 3.44 10 6 Mg 1.076 10 6 Si 1 10 6 S 5.15 10 5 Ar 1.01 10 5 Fe 9.00
nm (T = K, p = kP a (1atm( )), 1bar = 10 5 P a = atm) 1 ( ) m / m
 .1 1nm (T = 73.15K, p = 101.35kP a (1atm( )), 1bar = 10 5 P a = 0.9863atm) 1 ( ).413968 10 3 m 3 1 37. 1/3 3.34.414 10 3 m 3 6.0 10 3 = 3.7 (109 ) 3 (nm) 3 10 6 = 3.7 10 1 (nm) 3 = (3.34nm) 3 ( P = nrt,
.1 1nm (T = 73.15K, p = 101.35kP a (1atm( )), 1bar = 10 5 P a = 0.9863atm) 1 ( ).413968 10 3 m 3 1 37. 1/3 3.34.414 10 3 m 3 6.0 10 3 = 3.7 (109 ) 3 (nm) 3 10 6 = 3.7 10 1 (nm) 3 = (3.34nm) 3 ( P = nrt,
成長機構
 j im πmkt jin jim π mkt j q out j q im π mkt jin j j q out out π mkt π mkt dn dt πmkt dn v( ) Rmax bf dt πmkt R v ( J J ), J J, J J + + T T, J J m + Q+ / kt Q / kt + ( Q Q+ )/ ktm l / ktm J / J, l Q Q
j im πmkt jin jim π mkt j q out j q im π mkt jin j j q out out π mkt π mkt dn dt πmkt dn v( ) Rmax bf dt πmkt R v ( J J ), J J, J J + + T T, J J m + Q+ / kt Q / kt + ( Q Q+ )/ ktm l / ktm J / J, l Q Q
all.dvi
 72 9 Hooke,,,. Hooke. 9.1 Hooke 1 Hooke. 1, 1 Hooke. σ, ε, Young. σ ε (9.1), Young. τ γ G τ Gγ (9.2) X 1, X 2. Poisson, Poisson ν. ν ε 22 (9.) ε 11 F F X 2 X 1 9.1: Poisson 9.1. Hooke 7 Young Poisson G
72 9 Hooke,,,. Hooke. 9.1 Hooke 1 Hooke. 1, 1 Hooke. σ, ε, Young. σ ε (9.1), Young. τ γ G τ Gγ (9.2) X 1, X 2. Poisson, Poisson ν. ν ε 22 (9.) ε 11 F F X 2 X 1 9.1: Poisson 9.1. Hooke 7 Young Poisson G
2,200 WEB * Ξ ( ) η ( ) DC 1.5 i
 mailto: tomita@phys.h.kyoto-u.ac.jp 2002 2003 1.5 2003 2004 2005 2,200 WEB * Ξ ( ) η ( ) DC 1.5 i 1 1 1.1............................. 1 1.2..................................... 2 1.3.................................
mailto: tomita@phys.h.kyoto-u.ac.jp 2002 2003 1.5 2003 2004 2005 2,200 WEB * Ξ ( ) η ( ) DC 1.5 i 1 1 1.1............................. 1 1.2..................................... 2 1.3.................................
W u = u(x, t) u tt = a 2 u xx, a > 0 (1) D := {(x, t) : 0 x l, t 0} u (0, t) = 0, u (l, t) = 0, t 0 (2)
 3 215 4 27 1 1 u u(x, t) u tt a 2 u xx, a > (1) D : {(x, t) : x, t } u (, t), u (, t), t (2) u(x, ) f(x), u(x, ) t 2, x (3) u(x, t) X(x)T (t) u (1) 1 T (t) a 2 T (t) X (x) X(x) α (2) T (t) αa 2 T (t) (4)
3 215 4 27 1 1 u u(x, t) u tt a 2 u xx, a > (1) D : {(x, t) : x, t } u (, t), u (, t), t (2) u(x, ) f(x), u(x, ) t 2, x (3) u(x, t) X(x)T (t) u (1) 1 T (t) a 2 T (t) X (x) X(x) α (2) T (t) αa 2 T (t) (4)
untitled
 1907(40) 100 100 21 1 1 21 21 21 21 21 1891(M24) 1920(T9)1 2 1932(S7) 1947(S22) 1968(S43)12 2001(H13) 1955(S30) 1 24 9 7 22 43 13 2 100 6 200300t 1896M29 3876 1536 1 15 1907(M7) 11 6 1898(M31) 5 6 2 1912(M45)
1907(40) 100 100 21 1 1 21 21 21 21 21 1891(M24) 1920(T9)1 2 1932(S7) 1947(S22) 1968(S43)12 2001(H13) 1955(S30) 1 24 9 7 22 43 13 2 100 6 200300t 1896M29 3876 1536 1 15 1907(M7) 11 6 1898(M31) 5 6 2 1912(M45)
2 G(k) e ikx = (ik) n x n n! n=0 (k ) ( ) X n = ( i) n n k n G(k) k=0 F (k) ln G(k) = ln e ikx n κ n F (k) = F (k) (ik) n n= n! κ n κ n = ( i) n n k n
 . X {x, x 2, x 3,... x n } X X {, 2, 3, 4, 5, 6} X x i P i. 0 P i 2. n P i = 3. P (i ω) = i ω P i P 3 {x, x 2, x 3,... x n } ω P i = 6 X f(x) f(x) X n n f(x i )P i n x n i P i X n 2 G(k) e ikx = (ik) n
. X {x, x 2, x 3,... x n } X X {, 2, 3, 4, 5, 6} X x i P i. 0 P i 2. n P i = 3. P (i ω) = i ω P i P 3 {x, x 2, x 3,... x n } ω P i = 6 X f(x) f(x) X n n f(x i )P i n x n i P i X n 2 G(k) e ikx = (ik) n
[ ] (Ising model) 2 i S i S i = 1 (up spin : ) = 1 (down spin : ) (4.38) s z = ±1 4 H 0 = J zn/2 i,j S i S j (4.39) i, j z 5 2 z = 4 z = 6 3
![[ ] (Ising model) 2 i S i S i = 1 (up spin : ) = 1 (down spin : ) (4.38) s z = ±1 4 H 0 = J zn/2 i,j S i S j (4.39) i, j z 5 2 z = 4 z = 6 3 [ ] (Ising model) 2 i S i S i = 1 (up spin : ) = 1 (down spin : ) (4.38) s z = ±1 4 H 0 = J zn/2 i,j S i S j (4.39) i, j z 5 2 z = 4 z = 6 3](/thumbs/103/161261933.jpg) 4.2 4.2.1 [ ] (Ising model) 2 i S i S i = 1 (up spin : ) = 1 (down spin : ) (4.38) s z = ±1 4 H 0 = J zn/2 S i S j (4.39) i, j z 5 2 z = 4 z = 6 3 z = 6 z = 8 zn/2 1 2 N i z nearest neighbors of i j=1
4.2 4.2.1 [ ] (Ising model) 2 i S i S i = 1 (up spin : ) = 1 (down spin : ) (4.38) s z = ±1 4 H 0 = J zn/2 S i S j (4.39) i, j z 5 2 z = 4 z = 6 3 z = 6 z = 8 zn/2 1 2 N i z nearest neighbors of i j=1
ω 0 m(ẍ + γẋ + ω0x) 2 = ee (2.118) e iωt x = e 1 m ω0 2 E(ω). (2.119) ω2 iωγ Z N P(ω) = χ(ω)e = exzn (2.120) ϵ = ϵ 0 (1 + χ) ϵ(ω) ϵ 0 = 1 +
 2.6 2.6.1 ω 0 m(ẍ + γẋ + ω0x) 2 = ee (2.118) e iωt x = e 1 m ω0 2 E(ω). (2.119) ω2 iωγ Z N P(ω) = χ(ω)e = exzn (2.120) ϵ = ϵ 0 (1 + χ) ϵ(ω) ϵ 0 = 1 + Ne2 m j f j ω 2 j ω2 iωγ j (2.121) Z ω ω j γ j f j
2.6 2.6.1 ω 0 m(ẍ + γẋ + ω0x) 2 = ee (2.118) e iωt x = e 1 m ω0 2 E(ω). (2.119) ω2 iωγ Z N P(ω) = χ(ω)e = exzn (2.120) ϵ = ϵ 0 (1 + χ) ϵ(ω) ϵ 0 = 1 + Ne2 m j f j ω 2 j ω2 iωγ j (2.121) Z ω ω j γ j f j
 25 7 18 1 1 1.1 v.s............................. 1 1.1.1.................................. 1 1.1.2................................. 1 1.1.3.................................. 3 1.2................... 3
25 7 18 1 1 1.1 v.s............................. 1 1.1.1.................................. 1 1.1.2................................. 1 1.1.3.................................. 3 1.2................... 3
IA
 IA 31 4 11 1 1 4 1.1 Planck.............................. 4 1. Bohr.................................... 5 1.3..................................... 6 8.1................................... 8....................................
IA 31 4 11 1 1 4 1.1 Planck.............................. 4 1. Bohr.................................... 5 1.3..................................... 6 8.1................................... 8....................................
微分積分 サンプルページ この本の定価 判型などは, 以下の URL からご覧いただけます. このサンプルページの内容は, 初版 1 刷発行時のものです.
 微分積分 サンプルページ この本の定価 判型などは, 以下の URL からご覧いただけます. ttp://www.morikita.co.jp/books/mid/00571 このサンプルページの内容は, 初版 1 刷発行時のものです. i ii 014 10 iii [note] 1 3 iv 4 5 3 6 4 x 0 sin x x 1 5 6 z = f(x, y) 1 y = f(x)
微分積分 サンプルページ この本の定価 判型などは, 以下の URL からご覧いただけます. ttp://www.morikita.co.jp/books/mid/00571 このサンプルページの内容は, 初版 1 刷発行時のものです. i ii 014 10 iii [note] 1 3 iv 4 5 3 6 4 x 0 sin x x 1 5 6 z = f(x, y) 1 y = f(x)
120 9 I I 1 I 2 I 1 I 2 ( a) ( b) ( c ) I I 2 I 1 I ( d) ( e) ( f ) 9.1: Ampère (c) (d) (e) S I 1 I 2 B ds = µ 0 ( I 1 I 2 ) I 1 I 2 B ds =0. I 1 I 2
 9 E B 9.1 9.1.1 Ampère Ampère Ampère s law B S µ 0 B ds = µ 0 j ds (9.1) S rot B = µ 0 j (9.2) S Ampère Biot-Savart oulomb Gauss Ampère rot B 0 Ampère µ 0 9.1 (a) (b) I B ds = µ 0 I. I 1 I 2 B ds = µ 0
9 E B 9.1 9.1.1 Ampère Ampère Ampère s law B S µ 0 B ds = µ 0 j ds (9.1) S rot B = µ 0 j (9.2) S Ampère Biot-Savart oulomb Gauss Ampère rot B 0 Ampère µ 0 9.1 (a) (b) I B ds = µ 0 I. I 1 I 2 B ds = µ 0
II ( ) (7/31) II ( [ (3.4)] Navier Stokes [ (6/29)] Navier Stokes 3 [ (6/19)] Re
![II ( ) (7/31) II ( [ (3.4)] Navier Stokes [ (6/29)] Navier Stokes 3 [ (6/19)] Re II ( ) (7/31) II ( [ (3.4)] Navier Stokes [ (6/29)] Navier Stokes 3 [ (6/19)] Re](/thumbs/94/118770263.jpg) II 29 7 29-7-27 ( ) (7/31) II (http://www.damp.tottori-u.ac.jp/~ooshida/edu/fluid/) [ (3.4)] Navier Stokes [ (6/29)] Navier Stokes 3 [ (6/19)] Reynolds [ (4.6), (45.8)] [ p.186] Navier Stokes I Euler Navier
II 29 7 29-7-27 ( ) (7/31) II (http://www.damp.tottori-u.ac.jp/~ooshida/edu/fluid/) [ (3.4)] Navier Stokes [ (6/29)] Navier Stokes 3 [ (6/19)] Reynolds [ (4.6), (45.8)] [ p.186] Navier Stokes I Euler Navier
8 300 mm 2.50 m/s L/s ( ) 1.13 kg/m MPa 240 C 5.00mm 120 kpa ( ) kg/s c p = 1.02kJ/kgK, R = 287J/kgK kPa, 17.0 C 118 C 870m 3 R = 287J
 26 1 22 10 1 2 3 4 5 6 30.0 cm 1.59 kg 110kPa, 42.1 C, 18.0m/s 107kPa c p =1.02kJ/kgK 278J/kgK 30.0 C, 250kPa (c p = 1.02kJ/kgK, R = 287J/kgK) 18.0 C m/s 16.9 C 320kPa 270 m/s C c p = 1.02kJ/kgK, R = 292J/kgK
26 1 22 10 1 2 3 4 5 6 30.0 cm 1.59 kg 110kPa, 42.1 C, 18.0m/s 107kPa c p =1.02kJ/kgK 278J/kgK 30.0 C, 250kPa (c p = 1.02kJ/kgK, R = 287J/kgK) 18.0 C m/s 16.9 C 320kPa 270 m/s C c p = 1.02kJ/kgK, R = 292J/kgK
QMII_10.dvi
 65 1 1.1 1.1.1 1.1 H H () = E (), (1.1) H ν () = E ν () ν (). (1.) () () = δ, (1.3) μ () ν () = δ(μ ν). (1.4) E E ν () E () H 1.1: H α(t) = c (t) () + dνc ν (t) ν (), (1.5) H () () + dν ν () ν () = 1 (1.6)
65 1 1.1 1.1.1 1.1 H H () = E (), (1.1) H ν () = E ν () ν (). (1.) () () = δ, (1.3) μ () ν () = δ(μ ν). (1.4) E E ν () E () H 1.1: H α(t) = c (t) () + dνc ν (t) ν (), (1.5) H () () + dν ν () ν () = 1 (1.6)
The Physics of Atmospheres CAPTER :
 The Physics of Atmospheres CAPTER 4 1 4 2 41 : 2 42 14 43 17 44 25 45 27 46 3 47 31 48 32 49 34 41 35 411 36 maintex 23/11/28 The Physics of Atmospheres CAPTER 4 2 4 41 : 2 1 σ 2 (21) (22) k I = I exp(
The Physics of Atmospheres CAPTER 4 1 4 2 41 : 2 42 14 43 17 44 25 45 27 46 3 47 31 48 32 49 34 41 35 411 36 maintex 23/11/28 The Physics of Atmospheres CAPTER 4 2 4 41 : 2 1 σ 2 (21) (22) k I = I exp(
b3e2003.dvi
 15 II 5 5.1 (1) p, q p = (x + 2y, xy, 1), q = (x 2 + 3y 2, xyz, ) (i) p rotq (ii) p gradq D (2) a, b rot(a b) div [11, p.75] (3) (i) f f grad f = 1 2 grad( f 2) (ii) f f gradf 1 2 grad ( f 2) rotf 5.2
15 II 5 5.1 (1) p, q p = (x + 2y, xy, 1), q = (x 2 + 3y 2, xyz, ) (i) p rotq (ii) p gradq D (2) a, b rot(a b) div [11, p.75] (3) (i) f f grad f = 1 2 grad( f 2) (ii) f f gradf 1 2 grad ( f 2) rotf 5.2
: 2005 ( ρ t +dv j =0 r m m r = e E( r +e r B( r T 208 T = d E j 207 ρ t = = = e t δ( r r (t e r r δ( r r (t e r ( r δ( r r (t dv j =
 72 Maxwell. Maxwell e r ( =,,N Maxwell rot E + B t = 0 rot H D t = j dv D = ρ dv B = 0 D = ɛ 0 E H = μ 0 B ρ( r = j( r = N e δ( r r = N e r δ( r r = : 2005 ( 2006.8.22 73 207 ρ t +dv j =0 r m m r = e E(
72 Maxwell. Maxwell e r ( =,,N Maxwell rot E + B t = 0 rot H D t = j dv D = ρ dv B = 0 D = ɛ 0 E H = μ 0 B ρ( r = j( r = N e δ( r r = N e r δ( r r = : 2005 ( 2006.8.22 73 207 ρ t +dv j =0 r m m r = e E(
総研大恒星進化概要.dvi
 The Structure and Evolution of Stars I. Basic Equations. M r r =4πr2 ρ () P r = GM rρ. r 2 (2) r: M r : P and ρ: G: M r Lagrange r = M r 4πr 2 rho ( ) P = GM r M r 4πr. 4 (2 ) s(ρ, P ) s(ρ, P ) r L r T
The Structure and Evolution of Stars I. Basic Equations. M r r =4πr2 ρ () P r = GM rρ. r 2 (2) r: M r : P and ρ: G: M r Lagrange r = M r 4πr 2 rho ( ) P = GM r M r 4πr. 4 (2 ) s(ρ, P ) s(ρ, P ) r L r T
液晶の物理1:連続体理論(弾性,粘性)
 The Physics of Liquid Crystals P. G. de Gennes and J. Prost (Oxford University Press, 1993) Liquid crystals are beautiful and mysterious; I am fond of them for both reasons. My hope is that some readers
The Physics of Liquid Crystals P. G. de Gennes and J. Prost (Oxford University Press, 1993) Liquid crystals are beautiful and mysterious; I am fond of them for both reasons. My hope is that some readers
.2 ρ dv dt = ρk grad p + 3 η grad (divv) + η 2 v.3 divh = 0, rote + c H t = 0 dive = ρ, H = 0, E = ρ, roth c E t = c ρv E + H c t = 0 H c E t = c ρv T
 NHK 204 2 0 203 2 24 ( ) 7 00 7 50 203 2 25 ( ) 7 00 7 50 203 2 26 ( ) 7 00 7 50 203 2 27 ( ) 7 00 7 50 I. ( ν R n 2 ) m 2 n m, R = e 2 8πε 0 hca B =.09737 0 7 m ( ν = ) λ a B = 4πε 0ħ 2 m e e 2 = 5.2977
NHK 204 2 0 203 2 24 ( ) 7 00 7 50 203 2 25 ( ) 7 00 7 50 203 2 26 ( ) 7 00 7 50 203 2 27 ( ) 7 00 7 50 I. ( ν R n 2 ) m 2 n m, R = e 2 8πε 0 hca B =.09737 0 7 m ( ν = ) λ a B = 4πε 0ħ 2 m e e 2 = 5.2977
,., 5., ,. 2.2,., x z. y,.,,,. du dt + α p x = 0 dw dt + α p z + g = 0 α dp dt + pγ dα dt = 0 α V dα dt = 0 (2.2.1), γ = c p /c
 29 2 1 2.1 2.1.1.,., 5.,. 2.1.1,. 2.2,., x z. y,.,,,. du dt + α p x = 0 dw dt + α p z + g = 0 α dp dt + pγ dα dt = 0 α V dα dt = 0 (2.2.1), γ = c p /c v., V = (u, w), = ( / x, / z). 30 2.1.1: 31., U p(z),
29 2 1 2.1 2.1.1.,., 5.,. 2.1.1,. 2.2,., x z. y,.,,,. du dt + α p x = 0 dw dt + α p z + g = 0 α dp dt + pγ dα dt = 0 α V dα dt = 0 (2.2.1), γ = c p /c v., V = (u, w), = ( / x, / z). 30 2.1.1: 31., U p(z),
(5) 75 (a) (b) ( 1 ) v ( 1 ) E E 1 v (a) ( 1 ) x E E (b) (a) (b)
 (5) 74 Re, bondar laer (Prandtl) Re z ω z = x (5) 75 (a) (b) ( 1 ) v ( 1 ) E E 1 v (a) ( 1 ) x E E (b) (a) (b) (5) 76 l V x ) 1/ 1 ( 1 1 1 δ δ = x Re x p V x t V l l (1-1) 1/ 1 δ δ δ δ = x Re p V x t V
(5) 74 Re, bondar laer (Prandtl) Re z ω z = x (5) 75 (a) (b) ( 1 ) v ( 1 ) E E 1 v (a) ( 1 ) x E E (b) (a) (b) (5) 76 l V x ) 1/ 1 ( 1 1 1 δ δ = x Re x p V x t V l l (1-1) 1/ 1 δ δ δ δ = x Re p V x t V
4. ϵ(ν, T ) = c 4 u(ν, T ) ϵ(ν, T ) T ν π4 Planck dx = 0 e x 1 15 U(T ) x 3 U(T ) = σt 4 Stefan-Boltzmann σ 2π5 k 4 15c 2 h 3 = W m 2 K 4 5.
 A 1. Boltzmann Planck u(ν, T )dν = 8πh ν 3 c 3 kt 1 dν h 6.63 10 34 J s Planck k 1.38 10 23 J K 1 Boltzmann u(ν, T ) T ν e hν c = 3 10 8 m s 1 2. Planck λ = c/ν Rayleigh-Jeans u(ν, T )dν = 8πν2 kt dν c
A 1. Boltzmann Planck u(ν, T )dν = 8πh ν 3 c 3 kt 1 dν h 6.63 10 34 J s Planck k 1.38 10 23 J K 1 Boltzmann u(ν, T ) T ν e hν c = 3 10 8 m s 1 2. Planck λ = c/ν Rayleigh-Jeans u(ν, T )dν = 8πν2 kt dν c
x E E E e i ω = t + ikx 0 k λ λ 2π k 2π/λ k ω/v v n v c/n k = nω c c ω/2π λ k 2πn/λ 2π/(λ/n) κ n n κ N n iκ k = Nω c iωt + inωx c iωt + i( n+ iκ ) ωx
 x E E E e i ω t + ikx k λ λ π k π/λ k ω/v v n v c/n k nω c c ω/π λ k πn/λ π/(λ/n) κ n n κ N n iκ k Nω c iωt + inωx c iωt + i( n+ iκ ) ωx c κω x c iω ( t nx c) E E e E e E e e κ e ωκx/c e iω(t nx/c) I I
x E E E e i ω t + ikx k λ λ π k π/λ k ω/v v n v c/n k nω c c ω/π λ k πn/λ π/(λ/n) κ n n κ N n iκ k Nω c iωt + inωx c iωt + i( n+ iκ ) ωx c κω x c iω ( t nx c) E E e E e E e e κ e ωκx/c e iω(t nx/c) I I
Note.tex 2008/09/19( )
 1 20 9 19 2 1 5 1.1........................ 5 1.2............................. 8 2 9 2.1............................. 9 2.2.............................. 10 3 13 3.1.............................. 13 3.2..................................
1 20 9 19 2 1 5 1.1........................ 5 1.2............................. 8 2 9 2.1............................. 9 2.2.............................. 10 3 13 3.1.............................. 13 3.2..................................
³ÎΨÏÀ
 2017 12 12 Makoto Nakashima 2017 12 12 1 / 22 2.1. C, D π- C, D. A 1, A 2 C A 1 A 2 C A 3, A 4 D A 1 A 2 D Makoto Nakashima 2017 12 12 2 / 22 . (,, L p - ). Makoto Nakashima 2017 12 12 3 / 22 . (,, L p
2017 12 12 Makoto Nakashima 2017 12 12 1 / 22 2.1. C, D π- C, D. A 1, A 2 C A 1 A 2 C A 3, A 4 D A 1 A 2 D Makoto Nakashima 2017 12 12 2 / 22 . (,, L p - ). Makoto Nakashima 2017 12 12 3 / 22 . (,, L p
1
 1 1.. ( ) ( ) ( ) (A) E icb φ Et = + cdiva ct (H3) (B) ( ) ct ct ' ctct ' + ' = ' ct ' + ct ' i( ') (H3,H18) 3 (i) cosh Ψ = cosh ΘcoshΩ sinhψ sinhθ sinhω cosh Ψ cosh Θ cosh Ω = sinhψ sinhθ sinhω tanhψ
1 1.. ( ) ( ) ( ) (A) E icb φ Et = + cdiva ct (H3) (B) ( ) ct ct ' ctct ' + ' = ' ct ' + ct ' i( ') (H3,H18) 3 (i) cosh Ψ = cosh ΘcoshΩ sinhψ sinhθ sinhω cosh Ψ cosh Θ cosh Ω = sinhψ sinhθ sinhω tanhψ
現代物理化学 2-1(9)16.ppt
 --- S A, G U S S ds = d 'Q r / ΔS = S S = ds =,r,r d 'Q r r S -- ds = d 'Q r / ΔS = S S = ds =,r,r d 'Q r r d Q r e = P e = P ΔS d 'Q / e (d'q / e ) --3,e Q W Q (> 0),e e ΔU = Q + W = (Q + Q ) + W = 0
--- S A, G U S S ds = d 'Q r / ΔS = S S = ds =,r,r d 'Q r r S -- ds = d 'Q r / ΔS = S S = ds =,r,r d 'Q r r d Q r e = P e = P ΔS d 'Q / e (d'q / e ) --3,e Q W Q (> 0),e e ΔU = Q + W = (Q + Q ) + W = 0
 006 11 8 0 3 1 5 1.1..................... 5 1......................... 6 1.3.................... 6 1.4.................. 8 1.5................... 8 1.6................... 10 1.6.1......................
006 11 8 0 3 1 5 1.1..................... 5 1......................... 6 1.3.................... 6 1.4.................. 8 1.5................... 8 1.6................... 10 1.6.1......................
KENZOU
 KENZOU 2008 8 2 3 2 3 2 2 4 2 4............................................... 2 4.2............................... 3 4.2........................................... 4 4.3..............................
KENZOU 2008 8 2 3 2 3 2 2 4 2 4............................................... 2 4.2............................... 3 4.2........................................... 4 4.3..............................
1 s 1 H(s 1 ) N s 1, s,, s N H({s 1,, s N }) = N H(s k ) k=1 Z N =Tr {s1,,s N }e βh({s 1,,s N }) =Tr s1 Tr s Tr sn e β P k H(s k) N = Tr sk e βh(s k)
 19 1 14 007 3 1 1 Ising 4.1................................. 4................................... 5 3 9 3.1........................ 9 3................... 9 3.3........................ 11 4 14 4.1 Legendre..............................
19 1 14 007 3 1 1 Ising 4.1................................. 4................................... 5 3 9 3.1........................ 9 3................... 9 3.3........................ 11 4 14 4.1 Legendre..............................
 211 kotaro@math.titech.ac.jp 1 R *1 n n R n *2 R n = {(x 1,..., x n ) x 1,..., x n R}. R R 2 R 3 R n R n R n D D R n *3 ) (x 1,..., x n ) f(x 1,..., x n ) f D *4 n 2 n = 1 ( ) 1 f D R n f : D R 1.1. (x,
211 kotaro@math.titech.ac.jp 1 R *1 n n R n *2 R n = {(x 1,..., x n ) x 1,..., x n R}. R R 2 R 3 R n R n R n D D R n *3 ) (x 1,..., x n ) f(x 1,..., x n ) f D *4 n 2 n = 1 ( ) 1 f D R n f : D R 1.1. (x,
untitled
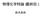 D nucleation 3 3D nucleation Glucose isomerase 10 V / nm s -1 5 0 0 5 10 C - C e / mg ml -1 kinetics µ R K kt kinetics kinetics kinetics r β π µ π r a r s + a s : β: µ πβ µ β s c s c a a r, & exp exp
D nucleation 3 3D nucleation Glucose isomerase 10 V / nm s -1 5 0 0 5 10 C - C e / mg ml -1 kinetics µ R K kt kinetics kinetics kinetics r β π µ π r a r s + a s : β: µ πβ µ β s c s c a a r, & exp exp
2 1 x 2 x 2 = RT 3πηaN A t (1.2) R/N A N A N A = N A m n(z) = n exp ( ) m gz k B T (1.3) z n z = m = m ρgv k B = erg K 1 R =
 1 1 1.1 1827 *1 195 *2 x 2 t x 2 = 2Dt D RT D = RT N A 1 6πaη (1.1) D N A a η 198 *3 ( a =.212µ) *1 Robert Brown (1773-1858. *2 Albert Einstein (1879-1955 *3 Jean Baptiste Perrin (187-1942 2 1 x 2 x 2
1 1 1.1 1827 *1 195 *2 x 2 t x 2 = 2Dt D RT D = RT N A 1 6πaη (1.1) D N A a η 198 *3 ( a =.212µ) *1 Robert Brown (1773-1858. *2 Albert Einstein (1879-1955 *3 Jean Baptiste Perrin (187-1942 2 1 x 2 x 2
( ; ) C. H. Scholz, The Mechanics of Earthquakes and Faulting : - ( ) σ = σ t sin 2π(r a) λ dσ d(r a) =
 1 9 8 1 1 1 ; 1 11 16 C. H. Scholz, The Mechanics of Earthquakes and Faulting 1. 1.1 1.1.1 : - σ = σ t sin πr a λ dσ dr a = E a = π λ σ πr a t cos λ 1 r a/λ 1 cos 1 E: σ t = Eλ πa a λ E/π γ : λ/ 3 γ =
1 9 8 1 1 1 ; 1 11 16 C. H. Scholz, The Mechanics of Earthquakes and Faulting 1. 1.1 1.1.1 : - σ = σ t sin πr a λ dσ dr a = E a = π λ σ πr a t cos λ 1 r a/λ 1 cos 1 E: σ t = Eλ πa a λ E/π γ : λ/ 3 γ =
理想気体ideal gasの熱力学的基本関係式
 the equipartition law of energy ( )kt k Boltzmann constant 5 Longman Dictionary of Physics (/)kt q Bq (/)kt equipartition law of energy mol (3/)kT (3/)RT (3/)R (5/)R 3R kt - equipartition of energy The
the equipartition law of energy ( )kt k Boltzmann constant 5 Longman Dictionary of Physics (/)kt q Bq (/)kt equipartition law of energy mol (3/)kT (3/)RT (3/)R (5/)R 3R kt - equipartition of energy The
1 (Berry,1975) 2-6 p (S πr 2 )p πr 2 p 2πRγ p p = 2γ R (2.5).1-1 : : : : ( ).2 α, β α, β () X S = X X α X β (.1) 1 2
 2005 9/8-11 2 2.2 ( 2-5) γ ( ) γ cos θ 2πr πρhr 2 g h = 2γ cos θ ρgr (2.1) γ = ρgrh (2.2) 2 cos θ θ cos θ = 1 (2.2) γ = 1 ρgrh (2.) 2 2. p p ρgh p ( ) p p = p ρgh (2.) h p p = 2γ r 1 1 (Berry,1975) 2-6
2005 9/8-11 2 2.2 ( 2-5) γ ( ) γ cos θ 2πr πρhr 2 g h = 2γ cos θ ρgr (2.1) γ = ρgrh (2.2) 2 cos θ θ cos θ = 1 (2.2) γ = 1 ρgrh (2.) 2 2. p p ρgh p ( ) p p = p ρgh (2.) h p p = 2γ r 1 1 (Berry,1975) 2-6
H.Haken Synergetics 2nd (1978)
 27 3 27 ) Ising Landau Synergetics Fokker-Planck F-P Landau F-P Gizburg-Landau G-L G-L Bénard/ Hopfield H.Haken Synergetics 2nd (1978) (1) Ising m T T C 1: m h Hamiltonian H = J ij S i S j h i S
27 3 27 ) Ising Landau Synergetics Fokker-Planck F-P Landau F-P Gizburg-Landau G-L G-L Bénard/ Hopfield H.Haken Synergetics 2nd (1978) (1) Ising m T T C 1: m h Hamiltonian H = J ij S i S j h i S
,, Andrej Gendiar (Density Matrix Renormalization Group, DMRG) 1 10 S.R. White [1, 2] 2 DMRG ( ) [3, 2] DMRG Baxter [4, 5] 2 Ising 2 1 Ising 1 1 Ising
![,, Andrej Gendiar (Density Matrix Renormalization Group, DMRG) 1 10 S.R. White [1, 2] 2 DMRG ( ) [3, 2] DMRG Baxter [4, 5] 2 Ising 2 1 Ising 1 1 Ising ,, Andrej Gendiar (Density Matrix Renormalization Group, DMRG) 1 10 S.R. White [1, 2] 2 DMRG ( ) [3, 2] DMRG Baxter [4, 5] 2 Ising 2 1 Ising 1 1 Ising](/thumbs/94/118770363.jpg) ,, Andrej Gendiar (Density Matrix Renormalization Group, DMRG) 1 10 S.R. White [1, 2] 2 DMRG ( ) [3, 2] DMRG Baxter [4, 5] 2 Ising 2 1 Ising 1 1 Ising Model 1 Ising 1 Ising Model N Ising (σ i = ±1) (Free
,, Andrej Gendiar (Density Matrix Renormalization Group, DMRG) 1 10 S.R. White [1, 2] 2 DMRG ( ) [3, 2] DMRG Baxter [4, 5] 2 Ising 2 1 Ising 1 1 Ising Model 1 Ising 1 Ising Model N Ising (σ i = ±1) (Free
Black-Scholes [1] Nelson [2] Schrödinger 1 Black Scholes [1] Black-Scholes Nelson [2][3][4] Schrödinger Nelson Parisi Wu [5] Nelson Parisi-W
![Black-Scholes [1] Nelson [2] Schrödinger 1 Black Scholes [1] Black-Scholes Nelson [2][3][4] Schrödinger Nelson Parisi Wu [5] Nelson Parisi-W Black-Scholes [1] Nelson [2] Schrödinger 1 Black Scholes [1] Black-Scholes Nelson [2][3][4] Schrödinger Nelson Parisi Wu [5] Nelson Parisi-W](/thumbs/97/132191371.jpg) 003 7 14 Black-Scholes [1] Nelson [] Schrödinger 1 Black Scholes [1] Black-Scholes Nelson [][3][4] Schrödinger Nelson Parisi Wu [5] Nelson Parisi-Wu Nelson e-mail: takatoshi-tasaki@nifty.com kabutaro@mocha.freemail.ne.jp
003 7 14 Black-Scholes [1] Nelson [] Schrödinger 1 Black Scholes [1] Black-Scholes Nelson [][3][4] Schrödinger Nelson Parisi Wu [5] Nelson Parisi-Wu Nelson e-mail: takatoshi-tasaki@nifty.com kabutaro@mocha.freemail.ne.jp
Einstein 1905 Lorentz Maxwell c E p E 2 (pc) 2 = m 2 c 4 (7.1) m E ( ) E p µ =(p 0,p 1,p 2,p 3 )=(p 0, p )= c, p (7.2) x µ =(x 0,x 1,x 2,x
 7 7.1 7.1.1 Einstein 1905 Lorentz Maxwell c E p E 2 (pc) 2 = m 2 c 4 (7.1) m E ( ) E p µ =(p 0,p 1,p 2,p 3 )=(p 0, p )= c, p (7.2) x µ =(x 0,x 1,x 2,x 3 )=(x 0, x )=(ct, x ) (7.3) E/c ct K = E mc 2 (7.4)
7 7.1 7.1.1 Einstein 1905 Lorentz Maxwell c E p E 2 (pc) 2 = m 2 c 4 (7.1) m E ( ) E p µ =(p 0,p 1,p 2,p 3 )=(p 0, p )= c, p (7.2) x µ =(x 0,x 1,x 2,x 3 )=(x 0, x )=(ct, x ) (7.3) E/c ct K = E mc 2 (7.4)
N/m f x x L dl U 1 du = T ds pdv + fdl (2.1)
 23 2 2.1 10 5 6 N/m 2 2.1.1 f x x L dl U 1 du = T ds pdv + fdl (2.1) 24 2 dv = 0 dl ( ) U f = T L p,t ( ) S L p,t (2.2) 2 ( ) ( ) S f = L T p,t p,l (2.3) ( ) U f = L p,t + T ( ) f T p,l (2.4) 1 f e ( U/
23 2 2.1 10 5 6 N/m 2 2.1.1 f x x L dl U 1 du = T ds pdv + fdl (2.1) 24 2 dv = 0 dl ( ) U f = T L p,t ( ) S L p,t (2.2) 2 ( ) ( ) S f = L T p,t p,l (2.3) ( ) U f = L p,t + T ( ) f T p,l (2.4) 1 f e ( U/
TOP URL 1
 TOP URL http://amonphys.web.fc.com/ 3.............................. 3.............................. 4.3 4................... 5.4........................ 6.5........................ 8.6...........................7
TOP URL http://amonphys.web.fc.com/ 3.............................. 3.............................. 4.3 4................... 5.4........................ 6.5........................ 8.6...........................7
m(ẍ + γẋ + ω 0 x) = ee (2.118) e iωt P(ω) = χ(ω)e = ex = e2 E(ω) m ω0 2 ω2 iωγ (2.119) Z N ϵ(ω) ϵ 0 = 1 + Ne2 m j f j ω 2 j ω2 iωγ j (2.120)
 2.6 2.6.1 mẍ + γẋ + ω 0 x) = ee 2.118) e iωt Pω) = χω)e = ex = e2 Eω) m ω0 2 ω2 iωγ 2.119) Z N ϵω) ϵ 0 = 1 + Ne2 m j f j ω 2 j ω2 iωγ j 2.120) Z ω ω j γ j f j f j f j sum j f j = Z 2.120 ω ω j, γ ϵω) ϵ
2.6 2.6.1 mẍ + γẋ + ω 0 x) = ee 2.118) e iωt Pω) = χω)e = ex = e2 Eω) m ω0 2 ω2 iωγ 2.119) Z N ϵω) ϵ 0 = 1 + Ne2 m j f j ω 2 j ω2 iωγ j 2.120) Z ω ω j γ j f j f j f j sum j f j = Z 2.120 ω ω j, γ ϵω) ϵ
18 2 F 12 r 2 r 1 (3) Coulomb km Coulomb M = kg F G = ( ) ( ) ( ) 2 = [N]. Coulomb
![18 2 F 12 r 2 r 1 (3) Coulomb km Coulomb M = kg F G = ( ) ( ) ( ) 2 = [N]. Coulomb 18 2 F 12 r 2 r 1 (3) Coulomb km Coulomb M = kg F G = ( ) ( ) ( ) 2 = [N]. Coulomb](/thumbs/93/114079295.jpg) r 1 r 2 r 1 r 2 2 Coulomb Gauss Coulomb 2.1 Coulomb 1 2 r 1 r 2 1 2 F 12 2 1 F 21 F 12 = F 21 = 1 4πε 0 1 2 r 1 r 2 2 r 1 r 2 r 1 r 2 (2.1) Coulomb ε 0 = 107 4πc 2 =8.854 187 817 10 12 C 2 N 1 m 2 (2.2)
r 1 r 2 r 1 r 2 2 Coulomb Gauss Coulomb 2.1 Coulomb 1 2 r 1 r 2 1 2 F 12 2 1 F 21 F 12 = F 21 = 1 4πε 0 1 2 r 1 r 2 2 r 1 r 2 r 1 r 2 (2.1) Coulomb ε 0 = 107 4πc 2 =8.854 187 817 10 12 C 2 N 1 m 2 (2.2)
chap03.dvi
 99 3 (Coriolis) cm m (free surface wave) 3.1 Φ 2.5 (2.25) Φ 100 3 r =(x, y, z) x y z F (x, y, z, t) =0 ( DF ) Dt = t + Φ F =0 onf =0. (3.1) n = F/ F (3.1) F n Φ = Φ n = 1 F F t Vn on F = 0 (3.2) Φ (3.1)
99 3 (Coriolis) cm m (free surface wave) 3.1 Φ 2.5 (2.25) Φ 100 3 r =(x, y, z) x y z F (x, y, z, t) =0 ( DF ) Dt = t + Φ F =0 onf =0. (3.1) n = F/ F (3.1) F n Φ = Φ n = 1 F F t Vn on F = 0 (3.2) Φ (3.1)
II 2 II
 II 2 II 2005 yugami@cc.utsunomiya-u.ac.jp 2005 4 1 1 2 5 2.1.................................... 5 2.2................................. 6 2.3............................. 6 2.4.................................
II 2 II 2005 yugami@cc.utsunomiya-u.ac.jp 2005 4 1 1 2 5 2.1.................................... 5 2.2................................. 6 2.3............................. 6 2.4.................................
) a + b = i + 6 b c = 6i j ) a = 0 b = c = 0 ) â = i + j 0 ˆb = 4) a b = b c = j + ) cos α = cos β = 6) a ˆb = b ĉ = 0 7) a b = 6i j b c = i + 6j + 8)
 4 4 ) a + b = i + 6 b c = 6i j ) a = 0 b = c = 0 ) â = i + j 0 ˆb = 4) a b = b c = j + ) cos α = cos β = 6) a ˆb = b ĉ = 0 7) a b = 6i j b c = i + 6j + 8) a b a b = 6i j 4 b c b c 9) a b = 4 a b) c = 7
4 4 ) a + b = i + 6 b c = 6i j ) a = 0 b = c = 0 ) â = i + j 0 ˆb = 4) a b = b c = j + ) cos α = cos β = 6) a ˆb = b ĉ = 0 7) a b = 6i j b c = i + 6j + 8) a b a b = 6i j 4 b c b c 9) a b = 4 a b) c = 7
68 A mm 1/10 A. (a) (b) A.: (a) A.3 A.4 1 1
 67 A Section A.1 0 1 0 1 Balmer 7 9 1 0.1 0.01 1 9 3 10:09 6 A.1: A.1 1 10 9 68 A 10 9 10 9 1 10 9 10 1 mm 1/10 A. (a) (b) A.: (a) A.3 A.4 1 1 A.1. 69 5 1 10 15 3 40 0 0 ¾ ¾ É f Á ½ j 30 A.3: A.4: 1/10
67 A Section A.1 0 1 0 1 Balmer 7 9 1 0.1 0.01 1 9 3 10:09 6 A.1: A.1 1 10 9 68 A 10 9 10 9 1 10 9 10 1 mm 1/10 A. (a) (b) A.: (a) A.3 A.4 1 1 A.1. 69 5 1 10 15 3 40 0 0 ¾ ¾ É f Á ½ j 30 A.3: A.4: 1/10
01_教職員.indd
 T. A. H. A. K. A. R. I. K. O. S. O. Y. O. M. K. Y. K. G. K. R. S. A. S. M. S. R. S. M. S. I. S. T. S. K.T. R. T. R. T. S. T. S. T. A. T. A. D. T. N. N. N. Y. N. S. N. S. H. R. H. W. H. T. H. K. M. K. M.
T. A. H. A. K. A. R. I. K. O. S. O. Y. O. M. K. Y. K. G. K. R. S. A. S. M. S. R. S. M. S. I. S. T. S. K.T. R. T. R. T. S. T. S. T. A. T. A. D. T. N. N. N. Y. N. S. N. S. H. R. H. W. H. T. H. K. M. K. M.
 2019 1 5 0 3 1 4 1.1.................... 4 1.1.1......................... 4 1.1.2........................ 5 1.1.3................... 5 1.1.4........................ 6 1.1.5......................... 6 1.2..........................
2019 1 5 0 3 1 4 1.1.................... 4 1.1.1......................... 4 1.1.2........................ 5 1.1.3................... 5 1.1.4........................ 6 1.1.5......................... 6 1.2..........................
H 0 H = H 0 + V (t), V (t) = gµ B S α qb e e iωt i t Ψ(t) = [H 0 + V (t)]ψ(t) Φ(t) Ψ(t) = e ih0t Φ(t) H 0 e ih0t Φ(t) + ie ih0t t Φ(t) = [
![H 0 H = H 0 + V (t), V (t) = gµ B S α qb e e iωt i t Ψ(t) = [H 0 + V (t)]ψ(t) Φ(t) Ψ(t) = e ih0t Φ(t) H 0 e ih0t Φ(t) + ie ih0t t Φ(t) = [ H 0 H = H 0 + V (t), V (t) = gµ B S α qb e e iωt i t Ψ(t) = [H 0 + V (t)]ψ(t) Φ(t) Ψ(t) = e ih0t Φ(t) H 0 e ih0t Φ(t) + ie ih0t t Φ(t) = [](/thumbs/99/141438442.jpg) 3 3. 3.. H H = H + V (t), V (t) = gµ B α B e e iωt i t Ψ(t) = [H + V (t)]ψ(t) Φ(t) Ψ(t) = e iht Φ(t) H e iht Φ(t) + ie iht t Φ(t) = [H + V (t)]e iht Φ(t) Φ(t) i t Φ(t) = V H(t)Φ(t), V H (t) = e iht V (t)e
3 3. 3.. H H = H + V (t), V (t) = gµ B α B e e iωt i t Ψ(t) = [H + V (t)]ψ(t) Φ(t) Ψ(t) = e iht Φ(t) H e iht Φ(t) + ie iht t Φ(t) = [H + V (t)]e iht Φ(t) Φ(t) i t Φ(t) = V H(t)Φ(t), V H (t) = e iht V (t)e
5 5.1 E 1, E 2 N 1, N 2 E tot N tot E tot = E 1 + E 2, N tot = N 1 + N 2 S 1 (E 1, N 1 ), S 2 (E 2, N 2 ) E 1, E 2 S tot = S 1 + S 2 2 S 1 E 1 = S 2 E
 5 5.1 E 1, E 2 N 1, N 2 E tot N tot E tot = E 1 + E 2, N tot = N 1 + N 2 S 1 (E 1, N 1 ), S 2 (E 2, N 2 ) E 1, E 2 S tot = S 1 + S 2 2 S 1 E 1 = S 2 E 2, S 1 N 1 = S 2 N 2 2 (chemical potential) µ S N
5 5.1 E 1, E 2 N 1, N 2 E tot N tot E tot = E 1 + E 2, N tot = N 1 + N 2 S 1 (E 1, N 1 ), S 2 (E 2, N 2 ) E 1, E 2 S tot = S 1 + S 2 2 S 1 E 1 = S 2 E 2, S 1 N 1 = S 2 N 2 2 (chemical potential) µ S N
 http://www.ike-dyn.ritsumei.ac.jp/ hyoo/wave.html 1 1, 5 3 1.1 1..................................... 3 1.2 5.1................................... 4 1.3.......................... 5 1.4 5.2, 5.3....................
http://www.ike-dyn.ritsumei.ac.jp/ hyoo/wave.html 1 1, 5 3 1.1 1..................................... 3 1.2 5.1................................... 4 1.3.......................... 5 1.4 5.2, 5.3....................
Hanbury-Brown Twiss (ver. 2.0) van Cittert - Zernike mutual coherence
 Hanbury-Brown Twiss (ver. 2.) 25 4 4 1 2 2 2 2.1 van Cittert - Zernike..................................... 2 2.2 mutual coherence................................. 4 3 Hanbury-Brown Twiss ( ) 5 3.1............................................
Hanbury-Brown Twiss (ver. 2.) 25 4 4 1 2 2 2 2.1 van Cittert - Zernike..................................... 2 2.2 mutual coherence................................. 4 3 Hanbury-Brown Twiss ( ) 5 3.1............................................
2 A B A B A A B Ea 1 51 Ea 1 A B A B B A B B A Ea 2 A B Ea 1 ( )k 1 Ea 1 Ea 2 Arrhenius 53 Ea R T k 1 = χe 1 Ea RT k 2 = χe 2 Ea RT 53 A B A B
 5. A B B A B A B B A A B A B 2 A [A] B [B] 51 v = k[a][b] 51 A B 3 0 273.16 A B A B A B A A [A] 52 v= k[a] 52 A B 55 2 A B A B A A B Ea 1 51 Ea 1 A B A B B A B B A Ea 2 A B Ea 1 ( )k 1 Ea 1 Ea 2 Arrhenius
5. A B B A B A B B A A B A B 2 A [A] B [B] 51 v = k[a][b] 51 A B 3 0 273.16 A B A B A B A A [A] 52 v= k[a] 52 A B 55 2 A B A B A A B Ea 1 51 Ea 1 A B A B B A B B A Ea 2 A B Ea 1 ( )k 1 Ea 1 Ea 2 Arrhenius
Onsager SOLUTION OF THE EIGENWERT PROBLEM (O-29) V = e H A e H B λ max Z 2 Onsager (O-77) (O-82) (O-83) Kramers-Wannier 1 1 Ons
 Onsager 2 9 207.2.7 3 SOLUTION OF THE EIGENWERT PROBLEM O-29 V = e H A e H B λ max Z 2 OnsagerO-77O-82 O-83 2 Kramers-Wannier Onsager * * * * * V self-adjoint V = V /2 V V /2 = V /2 V 2 V /2 = 2 sinh 2H
Onsager 2 9 207.2.7 3 SOLUTION OF THE EIGENWERT PROBLEM O-29 V = e H A e H B λ max Z 2 OnsagerO-77O-82 O-83 2 Kramers-Wannier Onsager * * * * * V self-adjoint V = V /2 V V /2 = V /2 V 2 V /2 = 2 sinh 2H
[ ] 0.1 lim x 0 e 3x 1 x IC ( 11) ( s114901) 0.2 (1) y = e 2x (x 2 + 1) (2) y = x/(x 2 + 1) 0.3 dx (1) 1 4x 2 (2) e x sin 2xdx (3) sin 2 xdx ( 11) ( s
![[ ] 0.1 lim x 0 e 3x 1 x IC ( 11) ( s114901) 0.2 (1) y = e 2x (x 2 + 1) (2) y = x/(x 2 + 1) 0.3 dx (1) 1 4x 2 (2) e x sin 2xdx (3) sin 2 xdx ( 11) ( s [ ] 0.1 lim x 0 e 3x 1 x IC ( 11) ( s114901) 0.2 (1) y = e 2x (x 2 + 1) (2) y = x/(x 2 + 1) 0.3 dx (1) 1 4x 2 (2) e x sin 2xdx (3) sin 2 xdx ( 11) ( s](/thumbs/94/118579915.jpg) [ ]. lim e 3 IC ) s49). y = e + ) ) y = / + ).3 d 4 ) e sin d 3) sin d ) s49) s493).4 z = y z z y s494).5 + y = 4 =.6 s495) dy = 3e ) d dy d = y s496).7 lim ) lim e s49).8 y = e sin ) y = sin e 3) y =
[ ]. lim e 3 IC ) s49). y = e + ) ) y = / + ).3 d 4 ) e sin d 3) sin d ) s49) s493).4 z = y z z y s494).5 + y = 4 =.6 s495) dy = 3e ) d dy d = y s496).7 lim ) lim e s49).8 y = e sin ) y = sin e 3) y =
1 filename=mathformula tex 1 ax 2 + bx + c = 0, x = b ± b 2 4ac, (1.1) 2a x 1 + x 2 = b a, x 1x 2 = c a, (1.2) ax 2 + 2b x + c = 0, x = b ± b 2
 filename=mathformula58.tex ax + bx + c =, x = b ± b 4ac, (.) a x + x = b a, x x = c a, (.) ax + b x + c =, x = b ± b ac. a (.3). sin(a ± B) = sin A cos B ± cos A sin B, (.) cos(a ± B) = cos A cos B sin
filename=mathformula58.tex ax + bx + c =, x = b ± b 4ac, (.) a x + x = b a, x x = c a, (.) ax + b x + c =, x = b ± b ac. a (.3). sin(a ± B) = sin A cos B ± cos A sin B, (.) cos(a ± B) = cos A cos B sin
第3章
 5 5.. Maxwell Maxwell-Ampere E H D P J D roth = J+ = J+ E+ P ( ε P = σe+ εe + (5. ( NL P= ε χe+ P NL, J = σe (5. Faraday rot = µ H E (5. (5. (5. ( E ( roth rot rot = µ NL µσ E µε µ P E (5.4 = ( = grad
5 5.. Maxwell Maxwell-Ampere E H D P J D roth = J+ = J+ E+ P ( ε P = σe+ εe + (5. ( NL P= ε χe+ P NL, J = σe (5. Faraday rot = µ H E (5. (5. (5. ( E ( roth rot rot = µ NL µσ E µε µ P E (5.4 = ( = grad
1 2 1 a(=,incident particle A(target nucleus) b (projectile B( product nucleus, residual nucleus, ) ; a + A B + b a A B b 1: A(a,b)B A=B,a=b 2 1. ( 10
 1 2 1 a(=,incident particle A(target nucleus) b (projectile B( product nucleus, residual nucleus, ) ; a + A B + b a A B b 1: A(a,b)B A=B,a=b 2 1. ( 10 14 m) ( 10 10 m) 2., 3 1 =reaction-text20181101b.tex
1 2 1 a(=,incident particle A(target nucleus) b (projectile B( product nucleus, residual nucleus, ) ; a + A B + b a A B b 1: A(a,b)B A=B,a=b 2 1. ( 10 14 m) ( 10 10 m) 2., 3 1 =reaction-text20181101b.tex
1 I 1.1 ± e = = - = C C MKSA [m], [Kg] [s] [A] 1C 1A 1 MKSA 1C 1C +q q +q q 1
![1 I 1.1 ± e = = - = C C MKSA [m], [Kg] [s] [A] 1C 1A 1 MKSA 1C 1C +q q +q q 1 1 I 1.1 ± e = = - = C C MKSA [m], [Kg] [s] [A] 1C 1A 1 MKSA 1C 1C +q q +q q 1](/thumbs/94/121802164.jpg) 1 I 1.1 ± e = = - =1.602 10 19 C C MKA [m], [Kg] [s] [A] 1C 1A 1 MKA 1C 1C +q q +q q 1 1.1 r 1,2 q 1, q 2 r 12 2 q 1, q 2 2 F 12 = k q 1q 2 r 12 2 (1.1) k 2 k 2 ( r 1 r 2 ) ( r 2 r 1 ) q 1 q 2 (q 1 q 2
1 I 1.1 ± e = = - =1.602 10 19 C C MKA [m], [Kg] [s] [A] 1C 1A 1 MKA 1C 1C +q q +q q 1 1.1 r 1,2 q 1, q 2 r 12 2 q 1, q 2 2 F 12 = k q 1q 2 r 12 2 (1.1) k 2 k 2 ( r 1 r 2 ) ( r 2 r 1 ) q 1 q 2 (q 1 q 2
Introduction to Numerical Analysis of Differential Equations Naoya Enomoto (Kyoto.univ.Dept.Science(math))
 Introduction to Numerical Analysis of Differential Equations Naoya Enomoto (Kyoto.univ.Dept.Science(math)) 2001 1 e-mail:s00x0427@ip.media.kyoto-u.ac.jp 1 1 Van der Pol 1 1 2 2 Bergers 2 KdV 2 1 5 1.1........................................
Introduction to Numerical Analysis of Differential Equations Naoya Enomoto (Kyoto.univ.Dept.Science(math)) 2001 1 e-mail:s00x0427@ip.media.kyoto-u.ac.jp 1 1 Van der Pol 1 1 2 2 Bergers 2 KdV 2 1 5 1.1........................................
1
 016 017 6 16 1 1 5 1.1............................................... 5 1................................................... 5 1.3................................................ 5 1.4...............................................
016 017 6 16 1 1 5 1.1............................................... 5 1................................................... 5 1.3................................................ 5 1.4...............................................
30 (11/04 )
 30 (11/04 ) i, 1,, II I?,,,,,,,,, ( ),,, ϵ δ,,,,, (, ),,,,,, 5 : (1) ( ) () (,, ) (3) ( ) (4) (5) ( ) (1),, (),,, () (3), (),, (4), (1), (3), ( ), (5),,,,,,,, ii,,,,,,,, Richard P. Feynman, The best teaching
30 (11/04 ) i, 1,, II I?,,,,,,,,, ( ),,, ϵ δ,,,,, (, ),,,,,, 5 : (1) ( ) () (,, ) (3) ( ) (4) (5) ( ) (1),, (),,, () (3), (),, (4), (1), (3), ( ), (5),,,,,,,, ii,,,,,,,, Richard P. Feynman, The best teaching
S I. dy fx x fx y fx + C 3 C dy fx 4 x, y dy v C xt y C v e kt k > xt yt gt [ v dt dt v e kt xt v e kt + C k x v + C C k xt v k 3 r r + dr e kt S dt d
 S I.. http://ayapin.film.s.dendai.ac.jp/~matuda /TeX/lecture.html PDF PS.................................... 3.3.................... 9.4................5.............. 3 5. Laplace................. 5....
S I.. http://ayapin.film.s.dendai.ac.jp/~matuda /TeX/lecture.html PDF PS.................................... 3.3.................... 9.4................5.............. 3 5. Laplace................. 5....
9 1. (Ti:Al 2 O 3 ) (DCM) (Cr:Al 2 O 3 ) (Cr:BeAl 2 O 4 ) Ĥ0 ψ n (r) ω n Schrödinger Ĥ 0 ψ n (r) = ω n ψ n (r), (1) ω i ψ (r, t) = [Ĥ0 + Ĥint (
 9 1. (Ti:Al 2 O 3 ) (DCM) (Cr:Al 2 O 3 ) (Cr:BeAl 2 O 4 ) 2. 2.1 Ĥ ψ n (r) ω n Schrödinger Ĥ ψ n (r) = ω n ψ n (r), (1) ω i ψ (r, t) = [Ĥ + Ĥint (t)] ψ (r, t), (2) Ĥ int (t) = eˆxe cos ωt ˆdE cos ωt, (3)
9 1. (Ti:Al 2 O 3 ) (DCM) (Cr:Al 2 O 3 ) (Cr:BeAl 2 O 4 ) 2. 2.1 Ĥ ψ n (r) ω n Schrödinger Ĥ ψ n (r) = ω n ψ n (r), (1) ω i ψ (r, t) = [Ĥ + Ĥint (t)] ψ (r, t), (2) Ĥ int (t) = eˆxe cos ωt ˆdE cos ωt, (3)
1: 3.3 1/8000 1/ m m/s v = 2kT/m = 2RT/M k R 8.31 J/(K mole) M 18 g 1 5 a v t πa 2 vt kg (
 1905 1 1.1 0.05 mm 1 µm 2 1 1 2004 21 2004 7 21 2005 web 2 [1, 2] 1 1: 3.3 1/8000 1/30 3 10 10 m 3 500 m/s 4 1 10 19 5 6 7 1.2 3 4 v = 2kT/m = 2RT/M k R 8.31 J/(K mole) M 18 g 1 5 a v t πa 2 vt 6 6 10
1905 1 1.1 0.05 mm 1 µm 2 1 1 2004 21 2004 7 21 2005 web 2 [1, 2] 1 1: 3.3 1/8000 1/30 3 10 10 m 3 500 m/s 4 1 10 19 5 6 7 1.2 3 4 v = 2kT/m = 2RT/M k R 8.31 J/(K mole) M 18 g 1 5 a v t πa 2 vt 6 6 10
80 4 r ˆρ i (r, t) δ(r x i (t)) (4.1) x i (t) ρ i ˆρ i t = 0 i r 0 t(> 0) j r 0 + r < δ(r 0 x i (0))δ(r 0 + r x j (t)) > (4.2) r r 0 G i j (r, t) dr 0
 79 4 4.1 4.1.1 x i (t) x j (t) O O r 0 + r r r 0 x i (0) r 0 x i (0) 4.1 L. van. Hove 1954 space-time correlation function V N 4.1 ρ 0 = N/V i t 80 4 r ˆρ i (r, t) δ(r x i (t)) (4.1) x i (t) ρ i ˆρ i t
79 4 4.1 4.1.1 x i (t) x j (t) O O r 0 + r r r 0 x i (0) r 0 x i (0) 4.1 L. van. Hove 1954 space-time correlation function V N 4.1 ρ 0 = N/V i t 80 4 r ˆρ i (r, t) δ(r x i (t)) (4.1) x i (t) ρ i ˆρ i t
i
 i 3 4 4 7 5 6 3 ( ).. () 3 () (3) (4) /. 3. 4/3 7. /e 8. a > a, a = /, > a >. () a >, a =, > a > () a > b, a = b, a < b. c c n a n + b n + c n 3c n..... () /3 () + (3) / (4) /4 (5) m > n, a b >, m > n,
i 3 4 4 7 5 6 3 ( ).. () 3 () (3) (4) /. 3. 4/3 7. /e 8. a > a, a = /, > a >. () a >, a =, > a > () a > b, a = b, a < b. c c n a n + b n + c n 3c n..... () /3 () + (3) / (4) /4 (5) m > n, a b >, m > n,
untitled
 40 4 4.3 I (1) I f (x) C [x 0 x 1 ] f (x) f 1 (x) (3.18) f (x) ; f 1 (x) = 1! f 00 ((x))(x ; x 0 )(x ; x 1 ) (x 0 (x) x 1 ) (4.8) (3.18) x (x) x 0 x 1 Z x1 f (x) dx ; Z x1 f 1 (x) dx = Z x1 = 1 1 Z x1
40 4 4.3 I (1) I f (x) C [x 0 x 1 ] f (x) f 1 (x) (3.18) f (x) ; f 1 (x) = 1! f 00 ((x))(x ; x 0 )(x ; x 1 ) (x 0 (x) x 1 ) (4.8) (3.18) x (x) x 0 x 1 Z x1 f (x) dx ; Z x1 f 1 (x) dx = Z x1 = 1 1 Z x1
) ] [ h m x + y + + V x) φ = Eφ 1) z E = i h t 13) x << 1) N n n= = N N + 1) 14) N n n= = N N + 1)N + 1) 6 15) N n 3 n= = 1 4 N N + 1) 16) N n 4
![) ] [ h m x + y + + V x) φ = Eφ 1) z E = i h t 13) x << 1) N n n= = N N + 1) 14) N n n= = N N + 1)N + 1) 6 15) N n 3 n= = 1 4 N N + 1) 16) N n 4 ) ] [ h m x + y + + V x) φ = Eφ 1) z E = i h t 13) x << 1) N n n= = N N + 1) 14) N n n= = N N + 1)N + 1) 6 15) N n 3 n= = 1 4 N N + 1) 16) N n 4](/thumbs/92/107866707.jpg) 1. k λ ν ω T v p v g k = π λ ω = πν = π T v p = λν = ω k v g = dω dk 1) ) 3) 4). p = hk = h λ 5) E = hν = hω 6) h = h π 7) h =6.6618 1 34 J sec) hc=197.3 MeV fm = 197.3 kev pm= 197.3 ev nm = 1.97 1 3 ev
1. k λ ν ω T v p v g k = π λ ω = πν = π T v p = λν = ω k v g = dω dk 1) ) 3) 4). p = hk = h λ 5) E = hν = hω 6) h = h π 7) h =6.6618 1 34 J sec) hc=197.3 MeV fm = 197.3 kev pm= 197.3 ev nm = 1.97 1 3 ev
all.dvi
 5,, Euclid.,..,... Euclid,.,.,, e i (i =,, ). 6 x a x e e e x.:,,. a,,. a a = a e + a e + a e = {e, e, e } a (.) = a i e i = a i e i (.) i= {a,a,a } T ( T ),.,,,,. (.),.,...,,. a 0 0 a = a 0 + a + a 0
5,, Euclid.,..,... Euclid,.,.,, e i (i =,, ). 6 x a x e e e x.:,,. a,,. a a = a e + a e + a e = {e, e, e } a (.) = a i e i = a i e i (.) i= {a,a,a } T ( T ),.,,,,. (.),.,...,,. a 0 0 a = a 0 + a + a 0
m d2 x = kx αẋ α > 0 (3.5 dt2 ( de dt = d dt ( 1 2 mẋ kx2 = mẍẋ + kxẋ = (mẍ + kxẋ = αẋẋ = αẋ 2 < 0 (3.6 Joule Joule 1843 Joule ( A B (> A ( 3-2
 3 3.1 ( 1 m d2 x(t dt 2 = kx(t k = (3.1 d 2 x dt 2 = ω2 x, ω = x(t = 0, ẋ(0 = v 0 k m (3.2 x = v 0 ω sin ωt (ẋ = v 0 cos ωt (3.3 E = 1 2 mẋ2 + 1 2 kx2 = 1 2 mv2 0 cos 2 ωt + 1 2 k v2 0 ω 2 sin2 ωt = 1
3 3.1 ( 1 m d2 x(t dt 2 = kx(t k = (3.1 d 2 x dt 2 = ω2 x, ω = x(t = 0, ẋ(0 = v 0 k m (3.2 x = v 0 ω sin ωt (ẋ = v 0 cos ωt (3.3 E = 1 2 mẋ2 + 1 2 kx2 = 1 2 mv2 0 cos 2 ωt + 1 2 k v2 0 ω 2 sin2 ωt = 1
医系の統計入門第 2 版 サンプルページ この本の定価 判型などは, 以下の URL からご覧いただけます. このサンプルページの内容は, 第 2 版 1 刷発行時のものです.
 医系の統計入門第 2 版 サンプルページ この本の定価 判型などは, 以下の URL からご覧いただけます. http://www.morikita.co.jp/books/mid/009192 このサンプルページの内容は, 第 2 版 1 刷発行時のものです. i 2 t 1. 2. 3 2 3. 6 4. 7 5. n 2 ν 6. 2 7. 2003 ii 2 2013 10 iii 1987
医系の統計入門第 2 版 サンプルページ この本の定価 判型などは, 以下の URL からご覧いただけます. http://www.morikita.co.jp/books/mid/009192 このサンプルページの内容は, 第 2 版 1 刷発行時のものです. i 2 t 1. 2. 3 2 3. 6 4. 7 5. n 2 ν 6. 2 7. 2003 ii 2 2013 10 iii 1987
 K E N Z U 2012 7 16 HP M. 1 1 4 1.1 3.......................... 4 1.2................................... 4 1.2.1..................................... 4 1.2.2.................................... 5................................
K E N Z U 2012 7 16 HP M. 1 1 4 1.1 3.......................... 4 1.2................................... 4 1.2.1..................................... 4 1.2.2.................................... 5................................
19 /
 19 / 1 1.1............................... 1. Andreev............................... 3 1.3..................................... 3 1.4..................................... 4 / 5.1......................................
19 / 1 1.1............................... 1. Andreev............................... 3 1.3..................................... 3 1.4..................................... 4 / 5.1......................................
 K E N Z U 01 7 16 HP M. 1 1 4 1.1 3.......................... 4 1.................................... 4 1..1..................................... 4 1...................................... 5................................
K E N Z U 01 7 16 HP M. 1 1 4 1.1 3.......................... 4 1.................................... 4 1..1..................................... 4 1...................................... 5................................







