|
|
|
- さみ ねぎたや
- 4 years ago
- Views:
Transcription
1
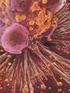
2 Pseudomonas aeruginosa S-827 Fig. 1. Method of biofilm formation. Table 1. Susceptibilities of antimicrobial agents on agar plate and in broth against Pseudomonas aeruginosa S-827
3 JULY CHEMOTHERAPY 888 Fig.2. Scanning electron microscopy 1992 of Pseudomonas aerugi- nosa S 827. (A) Fig. 4. Floating Bactericidal activity ceftazidime Fig. 3. Outer membrane profile of sessile and floating cells of Pseudornonas aeruginosa S827. Lanes: A, S-827; B, S-827 sessile cell (8 days); C, S-827 floating cell (8 days); D, molecular size standards were Cytochrome c hexamer (74.4 KDa), Cytochrome c tetramer (49.6 KDa), Cytochrome c trimer (37.2 KDa), and Cytochrome c dimer (24.4 KDa); E, S-827 sessile cell (3 days); F, S-827 floating cell (3 days). (A) in (-), cellお よ びsessile cellに 対 す る薬 剤 の殺 菌作 用 Floating TFLX,CPFXお ( ), and glucose. tosufloxacin 0.2ƒÊg/ml; 度 と し た Floating and (B) sessile Symbols: 0.2ƒÊg/ml; (ƒ ), ceft- cellに 対 し て キ ノロ ン は共 に 優 れ た殺 菌 力 を 示 し,薬 剤 接 触 後3時 で,両 薬 剤 共 生 菌 数 を1,000分 し め た そ れ に対 しCAZは せ え な か っ た ま た,sessile 剤2剤 quinolones aeruginosa cells 0.2% cells 6.25ƒÊg/ml. の 殺 菌 効 果 は,floating も の の,90%以 4.Floating and ciprofloxacin azidime 剤2剤 floating saline control: ( œ), 1MIC濃 of against Pseudomonas S-827 cells (B)Sessllc cells の1以 間 下 に まで減少 せ ま っ た く生 菌 数 を減 少 さ cellに 対 し て キ ノ ロ ン cellに 対 し て よ り劣 る 上 の 生 菌 数 の 減 少 が 認 め られ た しか し,CAZはfloating cellに 対 し て と同 様 ま っ た く効 果 を示 さ な か っ た cellあ る い はsessile よ びCAZの 示 し た 殺 菌 作 用 濃 度 は,3薬 cellに 対 す る 殺 菌 作 用 を,Fig.4に 剤 と も液 体 培 地 中 で の 5.TFLXと 多 糖 類 あ る い は タ ン パ ク分 解 酵 素 と の併 用効 果 0.2μg/mlのTFLXと,0.125mg/mlのlysozy-
4 Table 2. Activity of tosufloxacin combined with mucolytic enzymes against Pseudomonas aeruginosa S-827 sessile cells (A) Tosufloxacin (B) Ciprofloxacin (C) Ceftazidime Fig. 5. Bactericidal activity of (A) tosufloxacin, (B) ciprofloxacin, and (C) ceftazidime combined with erythromycin against Pseudomonas aeruginosa S-827 sessile cells in saline and 0.2% glucose. Symbols: (A): (-), control; ( ), tosufloxacin 0.2ƒÊg/ml;(ƒ ), erythromycin 200 ( œ), tosufloxacin 0.2 ƒêg/ml and erythromycin 200 (B): (-), control; ( ), ciprofloxacin 0.2ƒÊg/ml;
5 CHEMOTHERAPY JULY 1992 (A) Erythromycin (B) Clarithromycin Fig. 6. Bactericidal activity of tosufloxacin and (A) erythromycin or (B) clarithromycin against Pscudomonas acru,ginosa S 827 sessile cells in saline and 0.2% glucose. Symbols: (A): (-), control; ( œ), tosufloxacin 0.2ƒÊg/ ml, ( ), erythromycin 1ƒÊg/ml; ( œ), tosufloxacin 0.2 pg/m1 and erythromycin 1 g/ml. (B): (-), control; ( Z), tosufloxacin 0.2 pg/ml; (A), clarithromycin 1ƒÊg/ml; ( œ), tosufloxacin 0.2 pg/ml and clarithromycin 1 pg/nil. Fig. 7. Combined effect of tosufloxacin an erythromycin against Pseudomonas aeruginosa S-827 in brain heart infusion broth. Symbols: ( ), control; ( ), tosufloxacin 0.2 g/ml; (A), erythromycin 200 pg/ml; ( œ), ƒê tosufloxacin 0.2 pg/ml and erythromycin 200 g/ml. ƒê ƒê
6 VOL.40 NO.7
7 CHEMOTHERAPY JULY ) Buxton T B, Horner J, IIinton A, Rissing J P: In vivo glycocalyx expression by Staphylococcus aurcus phage type 52/52 A/80 in S. aurcus osteomyelitis. J Infect Dis 156: 942 `946, ) Da11 L, Barnes W G, Lane J W, Mills J: Enzymatic modification of glycocalyx in the treatment of experimental endocarditis due to viridans streptococci. J Infect Dis 156: 736 `740, ) Dix B A, Cohen P S, Laux D C, Cleeland R: Radiochemical method for evaluating the effect of antibiotics on Escherichia coli biofilms. Antimicrob Agents Chemother 32: 770 `772, 1988 activities of T 3262, a new fluoroquinolone. Antimicrob Agents Chemother 32: 827 `833, ) Goswami S K, Kivity S. Marom Z: Erythromycin inhibits respiratory glycoconjugate secretion 9) Anwar H, Costerton W: Enhanced activity of combination of tobramycin and piperacillin for eradication of sessile biofilm cells of Pseudonionas aeruginosa. Antimicrob Agents Chemother 34: 1666 `1671, ) Widmer A F, Wiestner A, Frei R, Zimmerli W: Killing of non-growing and adherent Escherichia coli determines drug efficacy in device-related infections. Antimicrob Agents Chemother 35: , 1991 ` 11) Fujimaki K, Noumi T, Saikawa I, Inoue M, Mitsuhashi S: In vitro and in vivo antibacterial from human airways in vitro, Am Rev Respir Dis 141: 72 `
8 VOL.40 NO.7 Characteristics of biofilm formed by Pseudornonas aeruginosa in vilro Kazuo Fujimaki, Yasushi Ikeda, Masahiro Takahata and Takashi Yasuda Research Laboratories, Toyama Chemical Co., Ltd., Shimookui, Toyama, Japan Biofilm formation by Pseudornonas aeruginosa was investigated by using medical teflon sheet as an artificial foreign device in vitro. Biofilm formed by the glycocalyx and cells was observed by scanning electron microscopy. Ceftazidime (CAZ) at 1 MIC did not show any bactericidal activity against sessile or floating cells. Tosufloxacin (TFLX) and ciprofloxacin (CPFX) had excellent bactericidal effects at 1 MIC against floating cells and a >90% decrease was observed in the number of viable sessile cells. Enhanced activities were observed against sessile cells not only with a combination of TFLX and CPFX at 1 MIC and erythromycin (EM) at 1/4 MIC to when the hourly changes of viable cells were observed but also for TFLX at 1 MIC and EM or clarithromycin at 1ƒÊg/ ml when the changes were observed over days. However, synergism was not observed when CAZ and EM were combined. The activity of TFLX was not enhanced when it was combined with lysozyme, hyaluronidase, protease or streptokinase.
CHEMOTHERAPY OCT. 1993
 CHEMOTHERAPY OCT. 1993 VOL. 41 NO. 10 CHEMOTHERAPY Table 1. Antibiotic permeability through biofilm of clinical isolates of Pseudomonas aeruginosa CAZ: ceftazidime, GM: gentamicin. Fig. 1. The effect of
CHEMOTHERAPY OCT. 1993 VOL. 41 NO. 10 CHEMOTHERAPY Table 1. Antibiotic permeability through biofilm of clinical isolates of Pseudomonas aeruginosa CAZ: ceftazidime, GM: gentamicin. Fig. 1. The effect of
 Table 1 Survival rates of infected mice given antibiotic doses producing peak serum a) S. aurcus Smith Challenge dose :7 ~10 (5% mucin) CFU/mouse. LD50: 1 ~103 (5% mucin) CFU/mouse. Table 2 Survival rates
Table 1 Survival rates of infected mice given antibiotic doses producing peak serum a) S. aurcus Smith Challenge dose :7 ~10 (5% mucin) CFU/mouse. LD50: 1 ~103 (5% mucin) CFU/mouse. Table 2 Survival rates
Fig.1 Chemical structure of BAY o 9867
 Fig.1 Chemical structure of BAY o 9867 CHEMOTHERAPY 43 Table 3 Antibacterial spectrum of gram negative bacteria Medium:Heart infusion agar (Nissui) Method:Agar dilution (Streak) CHEMOTHERAPY DEC 1985
Fig.1 Chemical structure of BAY o 9867 CHEMOTHERAPY 43 Table 3 Antibacterial spectrum of gram negative bacteria Medium:Heart infusion agar (Nissui) Method:Agar dilution (Streak) CHEMOTHERAPY DEC 1985
Table 1. MICs of fosfomycin and other antibiotics determined by agar dilution method against Pseudomonas aeruginosa FOM: fosfomycin, PIPC: piperacilli
 Key words: Pseudomonas aeruginosa, fosfomycin, ofloxacin, Table 1. MICs of fosfomycin and other antibiotics determined by agar dilution method against Pseudomonas aeruginosa FOM: fosfomycin, PIPC: piperacillin,
Key words: Pseudomonas aeruginosa, fosfomycin, ofloxacin, Table 1. MICs of fosfomycin and other antibiotics determined by agar dilution method against Pseudomonas aeruginosa FOM: fosfomycin, PIPC: piperacillin,
CHEMOTHERAPY APR Fig. 1 Chemical structure of cefotetan (CTT, YM09330)
 CHEMOTHERAPY APR. 1982 Fig. 1 Chemical structure of cefotetan (CTT, YM09330) VOL.30 S-1 CHEMOTHERAPY Fig. 2 Comparison of standard curves of CTT on various test organisms by cylinder plate method Column
CHEMOTHERAPY APR. 1982 Fig. 1 Chemical structure of cefotetan (CTT, YM09330) VOL.30 S-1 CHEMOTHERAPY Fig. 2 Comparison of standard curves of CTT on various test organisms by cylinder plate method Column
coccus aureus Corynebacterium sp, Haemophilus parainfluenzae Klebsiella pneumoniae Pseudornonas aeruginosa Pseudomonas sp., Xanthomonas maltophilia, F
 VOL.43 S-1 coccus aureus Corynebacterium sp, Haemophilus parainfluenzae Klebsiella pneumoniae Pseudornonas aeruginosa Pseudomonas sp., Xanthomonas maltophilia, Flavobacter- Table 1. Concentration of grepafloxacin
VOL.43 S-1 coccus aureus Corynebacterium sp, Haemophilus parainfluenzae Klebsiella pneumoniae Pseudornonas aeruginosa Pseudomonas sp., Xanthomonas maltophilia, Flavobacter- Table 1. Concentration of grepafloxacin
Clostridium difficile ciprofloxacin, ofloxacin, norfloxacin Bifidobacterium Lactobacillus Lactobacillus Bacteroides fragilis B. fragilis C. difficile
 Clostridium difficile ciprofloxacin, ofloxacin, norfloxacin Bifidobacterium Lactobacillus Lactobacillus Bacteroides fragilis B. fragilis C. difficile Key words: temafloxacin, TA-167, Bacteroides fragilis,
Clostridium difficile ciprofloxacin, ofloxacin, norfloxacin Bifidobacterium Lactobacillus Lactobacillus Bacteroides fragilis B. fragilis C. difficile Key words: temafloxacin, TA-167, Bacteroides fragilis,
CHEMOTHERAPY
 CHEMOTHERAPY VOL.41 S-2 Laboratory and clinical evaluation of teicoplanin CHEMOTHERAPY AUG. 1993 VOL.41 S-2 Laboratory and clinical evaluation of teicoplanin Table 1. Comparative in vitro activity of teicoplanin
CHEMOTHERAPY VOL.41 S-2 Laboratory and clinical evaluation of teicoplanin CHEMOTHERAPY AUG. 1993 VOL.41 S-2 Laboratory and clinical evaluation of teicoplanin Table 1. Comparative in vitro activity of teicoplanin
CHEMOTHERAPY FEB Table 1. Activity of cefpirome and others against clinical isolates
 VOL.39 S-1 CHEMOTHERAPY FEB. 1981 Table 1. Activity of cefpirome and others against clinical isolates VOL.39 S-1 CHEMOTHERAPY FEB. 1991 72 M, 55.5 kg 66 F, 53 kg Chronic bronchitis Bronchopneumonia Peak
VOL.39 S-1 CHEMOTHERAPY FEB. 1981 Table 1. Activity of cefpirome and others against clinical isolates VOL.39 S-1 CHEMOTHERAPY FEB. 1991 72 M, 55.5 kg 66 F, 53 kg Chronic bronchitis Bronchopneumonia Peak
CHEMOTHERAPY aureus 0.10, Enterococcus faecalis 3.13, Escherichia coli 0.20, Klebsiella pneumoniae, Enterobacter spp., Serratia marcescens 0.78, Prote
 aureus 0.10, Enterococcus faecalis 3.13, Escherichia coli 0.20, Klebsiella pneumoniae, Enterobacter spp., Serratia marcescens 0.78, Proteus mirabilis 3.13, Proteus vulgaris 1.56, Citrobacter freundii 0.39,
aureus 0.10, Enterococcus faecalis 3.13, Escherichia coli 0.20, Klebsiella pneumoniae, Enterobacter spp., Serratia marcescens 0.78, Proteus mirabilis 3.13, Proteus vulgaris 1.56, Citrobacter freundii 0.39,
 VOL.32 S-9 CHEMOTHERAPY Table 1 Minimum inhibitory concentrations of AC-1370, CPZ and CAZ Table 2 Efficacy of AC-1370 and CPZ against systemic infections in mice *Inoculum size: 106 cells/ml * 95% confidence
VOL.32 S-9 CHEMOTHERAPY Table 1 Minimum inhibitory concentrations of AC-1370, CPZ and CAZ Table 2 Efficacy of AC-1370 and CPZ against systemic infections in mice *Inoculum size: 106 cells/ml * 95% confidence
 CHEMOTHERAPY OCT. 1994 Tazobactam Piperacillin Fig. I. Chemical structures of tazobactam and piperacillin. Table 1. Media used for preculture and MIC determination BHIB: Brain heart infusion broth (Difco),
CHEMOTHERAPY OCT. 1994 Tazobactam Piperacillin Fig. I. Chemical structures of tazobactam and piperacillin. Table 1. Media used for preculture and MIC determination BHIB: Brain heart infusion broth (Difco),
 Table 1.Resistance criteria Fig.1.The resistance rates of piperacillin,ceftazidime, cefsulodin,imipenem,aztreonam,gentamicin,tobramycin,amikacin,isepamicin,fosfomycin and ofloxacin against 2,793 strains
Table 1.Resistance criteria Fig.1.The resistance rates of piperacillin,ceftazidime, cefsulodin,imipenem,aztreonam,gentamicin,tobramycin,amikacin,isepamicin,fosfomycin and ofloxacin against 2,793 strains
epidermidis, Enterococcus faecalis, Enterococcus Klebsiella pneumoniae, Proteus mirabilis, indolepositive Proteus spp., Enterobacter spp., Serratia
 epidermidis, Enterococcus faecalis, Enterococcus Klebsiella pneumoniae, Proteus mirabilis, indolepositive Proteus spp., Enterobacter spp., Serratia Table 3. Overall clinical efficacy of cefozopran in
epidermidis, Enterococcus faecalis, Enterococcus Klebsiella pneumoniae, Proteus mirabilis, indolepositive Proteus spp., Enterobacter spp., Serratia Table 3. Overall clinical efficacy of cefozopran in
VOL.32 S-7 CHEMOTHERAPY Table 1 MIC of standard strains of CTRX Fig. 2 Cumulative curves of MIC S. aureus (26 strains )
 CHEMOTHERAPY OCT. 1984 Fig. I Chemical structure of CTRX VOL.32 S-7 CHEMOTHERAPY Table 1 MIC of standard strains of CTRX Fig. 2 Cumulative curves of MIC S. aureus (26 strains ) CHEMOTHERAPY Fig. 3 Cumulative
CHEMOTHERAPY OCT. 1984 Fig. I Chemical structure of CTRX VOL.32 S-7 CHEMOTHERAPY Table 1 MIC of standard strains of CTRX Fig. 2 Cumulative curves of MIC S. aureus (26 strains ) CHEMOTHERAPY Fig. 3 Cumulative
1272 CHEMOTHERAPY MAR. 1975
 1272 CHEMOTHERAPY MAR. 1975 VOL. 23 NO. 3 CHEMOTHERAPY 1273 Fig. 2 Minimal inhibitory concentration of aminoglycosides against 50 strains of Klebsiella Fig. 1 Minimal inhibitory concentration of aminoglycosides
1272 CHEMOTHERAPY MAR. 1975 VOL. 23 NO. 3 CHEMOTHERAPY 1273 Fig. 2 Minimal inhibitory concentration of aminoglycosides against 50 strains of Klebsiella Fig. 1 Minimal inhibitory concentration of aminoglycosides
 Table 1 Antibacterial spectra of CPM and other antimicrobials against anaerobes Fig. 1 In vitro activity of CPM and other antibiotics against B. fragilis (136 strains) Fig. 2 In vitro activity of CPM and
Table 1 Antibacterial spectra of CPM and other antimicrobials against anaerobes Fig. 1 In vitro activity of CPM and other antibiotics against B. fragilis (136 strains) Fig. 2 In vitro activity of CPM and
CHEMOTHERAPY JUNE 1987 Table1 Media used *BHIB, brain heart infusion broth (Difco); /3 -NAD, S -nicotinamidoadeninedinucleotide (Sigma Chemical Co.);
 VOL.35 S-2 CHEMOTHERAPY Fig.1 Chemical structure of carumonam CHEMOTHERAPY JUNE 1987 Table1 Media used *BHIB, brain heart infusion broth (Difco); /3 -NAD, S -nicotinamidoadeninedinucleotide (Sigma Chemical
VOL.35 S-2 CHEMOTHERAPY Fig.1 Chemical structure of carumonam CHEMOTHERAPY JUNE 1987 Table1 Media used *BHIB, brain heart infusion broth (Difco); /3 -NAD, S -nicotinamidoadeninedinucleotide (Sigma Chemical
 Table 1.Concentration of gatifloxacin (Middle-ear) Table 2.Concentration of gatifloxacin (Paranasal sinuses) Table 3.Concentration of gatifloxacin (Tonsil) Table 4.No.of patients studied Table 5.Background
Table 1.Concentration of gatifloxacin (Middle-ear) Table 2.Concentration of gatifloxacin (Paranasal sinuses) Table 3.Concentration of gatifloxacin (Tonsil) Table 4.No.of patients studied Table 5.Background
VOL.35 S-2 CHEMOTHERAPY Table 1 Sex and age distribution Table 2 Applications of treatment with carumonam Table 3 Concentration of carumonam in human
 CHEMOTHERAPY Fig. 1 Chemical structure of carumonam Disodium(+)-(Z)-CCE1-(2-amino-4-thiazoly1)-2-[[(2S, -(carbamoyloxymethyl)-4-oxo-1-sulfonato-3-azetidinyll -2-oxoethylidene] amino] oxy] acetate 3S)-2
CHEMOTHERAPY Fig. 1 Chemical structure of carumonam Disodium(+)-(Z)-CCE1-(2-amino-4-thiazoly1)-2-[[(2S, -(carbamoyloxymethyl)-4-oxo-1-sulfonato-3-azetidinyll -2-oxoethylidene] amino] oxy] acetate 3S)-2
 CHEMOTHERAPY APRIL 1992 VOL. 40 S- 1 Table 1-1. Comparative in vitro activity of meropenem against clinical isolates CNS: coagulase-negative staphylococci CHEMOTHERAPY APRIL 1992 Table 1-2. Comparative
CHEMOTHERAPY APRIL 1992 VOL. 40 S- 1 Table 1-1. Comparative in vitro activity of meropenem against clinical isolates CNS: coagulase-negative staphylococci CHEMOTHERAPY APRIL 1992 Table 1-2. Comparative
VOL.47 NO.5 Table 1. Susceptibility distribution of Ĉ- lactams against clinical isolates of MRSA MRSA: rnethicillin- resistant Staphylococcus aureus
 MAY 1999 VOL.47 NO.5 Table 1. Susceptibility distribution of ƒà- lactams against clinical isolates of MRSA MRSA: rnethicillin- resistant Staphylococcus aureus (oxacillin MIC: 4ƒÊg/ ml) FMOX: flomoxef,
MAY 1999 VOL.47 NO.5 Table 1. Susceptibility distribution of ƒà- lactams against clinical isolates of MRSA MRSA: rnethicillin- resistant Staphylococcus aureus (oxacillin MIC: 4ƒÊg/ ml) FMOX: flomoxef,
Table 1. Antibacterial activitiy of grepafloxacin and other antibiotics against clinical isolates
 Table 1. Antibacterial activitiy of grepafloxacin and other antibiotics against clinical isolates Table 2-1. Summary of patients treated with grepafloxacin for respiratory infection 1) Out: outpatient,
Table 1. Antibacterial activitiy of grepafloxacin and other antibiotics against clinical isolates Table 2-1. Summary of patients treated with grepafloxacin for respiratory infection 1) Out: outpatient,
 b) Gram-negative bacteria Fig. 2 Sensitivity distribution of clinical isolates : E. coli Fig. 3 Sensitivity distribution of clinical isolates : Pseudomonas Fig. 1 Sensitivity distribution of clinical isolates
b) Gram-negative bacteria Fig. 2 Sensitivity distribution of clinical isolates : E. coli Fig. 3 Sensitivity distribution of clinical isolates : Pseudomonas Fig. 1 Sensitivity distribution of clinical isolates
VOL. 36 S-3 CHEMOTHERAPY 437
 VOL. 36 S-3 CHEMOTHERAPY 437 438 CHEMOTHERAPY JULY 1988 Fig. 1 Contractile response of gastrointestinal tract to intravenous administration of saline and EM in interdigestive state in dogs (a) : Saline,
VOL. 36 S-3 CHEMOTHERAPY 437 438 CHEMOTHERAPY JULY 1988 Fig. 1 Contractile response of gastrointestinal tract to intravenous administration of saline and EM in interdigestive state in dogs (a) : Saline,
Fig. 1 Chemical structure of DL-8280
 Fig. 1 Chemical structure of DL-8280 Fig. 2 Susceptibility of cl in ical isolates to DL4280 Fig. 5 Susceptibility of clinical isolates to DL-8280 Fig. 3 Susceptibility of clinical isolates to DL-8280 Fig.
Fig. 1 Chemical structure of DL-8280 Fig. 2 Susceptibility of cl in ical isolates to DL4280 Fig. 5 Susceptibility of clinical isolates to DL-8280 Fig. 3 Susceptibility of clinical isolates to DL-8280 Fig.
Staphylococcus sp. K.pneumoniae P.mirabilis C.freundii E. cloacae Serratia sp. P. aeruginosa ml, Enterococcus avium >100ƒÊg/ml
 CHEMOTHERAPY SEPT. 1992 cefoperazone ceftazidime (CAZ), imipenem (IPM) Staphylococcus sp., Enterococcus (CPZ), faecalis, Escherichia coli, Klebsiella pneumoniae, Citrobacter freundii, Enterobacter cloacae,
CHEMOTHERAPY SEPT. 1992 cefoperazone ceftazidime (CAZ), imipenem (IPM) Staphylococcus sp., Enterococcus (CPZ), faecalis, Escherichia coli, Klebsiella pneumoniae, Citrobacter freundii, Enterobacter cloacae,
CHEMOTHERAPY Fig. 1 Chemical structure of CXM-AX
 Fig. 1 Chemical structure of CXM-AX NOV. 1986 Fig. 2 Sensitivity distribution of clinical isolates organisms (106 cells/ml) a Smurcus 27 strains d) P.m irabilis 15 strains b Ecol i 27 strains 111.morganii
Fig. 1 Chemical structure of CXM-AX NOV. 1986 Fig. 2 Sensitivity distribution of clinical isolates organisms (106 cells/ml) a Smurcus 27 strains d) P.m irabilis 15 strains b Ecol i 27 strains 111.morganii
CHEMOTHERAPY 52 MAY d a a. Normal S. aureus d Đg/ml, 2h; e b b Đg/ml, Abnormal e. division lh; Abnormal thickening division cross f c. 1
 Levloxacin V.:1 Z iiiir fa L CHEMOTHERAPY 52 MAY d a a. Normal S. aureus d. 0.39 Đg/ml, 2h; e b b. 0.10 Đg/ml, Abnormal e. division lh; Abnormal thickening division cross f c. 1992 0.39 Đg/ml, and Fig.
Levloxacin V.:1 Z iiiir fa L CHEMOTHERAPY 52 MAY d a a. Normal S. aureus d. 0.39 Đg/ml, 2h; e b b. 0.10 Đg/ml, Abnormal e. division lh; Abnormal thickening division cross f c. 1992 0.39 Đg/ml, and Fig.
 VOL.30 NO.12 CHEMOTHERAPY Fig. 1 Effect of temperature on the growth curve of A. calcoaceticus A. calcoaceticits Ac 54 A. calcoacetictts Ac 164 Fig. 2 Effect of medium ph on the growth curve of A. calcoaceticus
VOL.30 NO.12 CHEMOTHERAPY Fig. 1 Effect of temperature on the growth curve of A. calcoaceticus A. calcoaceticits Ac 54 A. calcoacetictts Ac 164 Fig. 2 Effect of medium ph on the growth curve of A. calcoaceticus
CHEMOTHERAPY JUN Citrobacter freundii 27, Enterobacter aerogenes 26, Enterobacter cloacae 27, Proteus rettgeri 7, Proteus inconstans 20, Proteus
 VOL. 32 S-4 CHEMOTHERAPY Fig. 1 Chemical structure of sodium cefoperazone Fig. 2 Chemical structure of sodium cefoperazone CHEMOTHERAPY JUN. 1984 Citrobacter freundii 27, Enterobacter aerogenes 26, Enterobacter
VOL. 32 S-4 CHEMOTHERAPY Fig. 1 Chemical structure of sodium cefoperazone Fig. 2 Chemical structure of sodium cefoperazone CHEMOTHERAPY JUN. 1984 Citrobacter freundii 27, Enterobacter aerogenes 26, Enterobacter
CHEMOTHERAPY APRIL 1992 Acinetobacter calcoaceticus Staphylococcus aureus, Escherichia coli P. aeruginosa E. eoli, Klebsiella pneumoniae Serratia marc
 APRIL 1992 Acinetobacter calcoaceticus Staphylococcus aureus, Escherichia coli P. aeruginosa E. eoli, Klebsiella pneumoniae Serratia marcescens P. aeruginosa P. aeruginosa Streptococcus pyogenes Streptococcus
APRIL 1992 Acinetobacter calcoaceticus Staphylococcus aureus, Escherichia coli P. aeruginosa E. eoli, Klebsiella pneumoniae Serratia marcescens P. aeruginosa P. aeruginosa Streptococcus pyogenes Streptococcus
 Fig. 1. Structures of NM394, NAD-358 and NAD-245 Fig. 2. Typical HPLC chromatograms of NM394 in human plasma by organic solvent extraction method (a): Blank plasma (b): Plasma spiked with NM394 and internal
Fig. 1. Structures of NM394, NAD-358 and NAD-245 Fig. 2. Typical HPLC chromatograms of NM394 in human plasma by organic solvent extraction method (a): Blank plasma (b): Plasma spiked with NM394 and internal
CHEMOTHERAPY
 CHEMOTHERAPY CHEMOTHERAPY Table 1 Antibacterial activity of Sulbactam/CPZ against standard strains MIC mg/ml Inoculum size 106 CFU/ml * Sulbactam/CPZ= 1: 1 ** Concentration of Sulbactam+ CPZ CHEMOTHERAPY
CHEMOTHERAPY CHEMOTHERAPY Table 1 Antibacterial activity of Sulbactam/CPZ against standard strains MIC mg/ml Inoculum size 106 CFU/ml * Sulbactam/CPZ= 1: 1 ** Concentration of Sulbactam+ CPZ CHEMOTHERAPY
988 CHEMOTHERAPY NOV. 1971
 988 CHEMOTHERAPY NOV. 1971 VOL. 19 NO. 8 CHEMOTHERAPY 989 Effect of medium-ph and inoculum size on activity of SB-PC heart infusion agar, mcg/ml Sensitivity distribution of Staphylococci to SB-PC in surgical
988 CHEMOTHERAPY NOV. 1971 VOL. 19 NO. 8 CHEMOTHERAPY 989 Effect of medium-ph and inoculum size on activity of SB-PC heart infusion agar, mcg/ml Sensitivity distribution of Staphylococci to SB-PC in surgical
CHEMOTHERAPY Methicillin-resistant S.aureus(MRSA) coccus epidermidis 105 Streptococcus pyogenes E.faecali senterococcus avium Enterococcus faecium Str
 cefaclor(ccl),cefuroxime(cxm),cefixime (CFIX),cefteram(CFTM),cefdinir(CFDN) pneumoniae,streptococcus pyogenes Moraxella catarrhalis,haemophilus influenzae,escherichia coli, Klebsiella pneumoniae,proteus
cefaclor(ccl),cefuroxime(cxm),cefixime (CFIX),cefteram(CFTM),cefdinir(CFDN) pneumoniae,streptococcus pyogenes Moraxella catarrhalis,haemophilus influenzae,escherichia coli, Klebsiella pneumoniae,proteus
CHEMOTHERAPY NOV S. aureus, S. epidermidis, E. coli, K. pgeumoniae, E. cloacae, S. marcescens, P. mirabilis, Proteus, P. aeruginosa Inoculum siz
 VOL.33 S-5 CHEMOTHERAPY 381 Fig. 1 Chemical structure of HAPA-B Chemical name 1-N-[(2S)-3-Amino-2-hydroxypropiony1]-4-0-(6-amino- 6-deoxy-a-D-glucopyranosyl)-6-013-deoxy-4-C-methyl- 3-(methylamino)-ƒÀ-L-arabinopyranosyl]-2-deoxystreptamine
VOL.33 S-5 CHEMOTHERAPY 381 Fig. 1 Chemical structure of HAPA-B Chemical name 1-N-[(2S)-3-Amino-2-hydroxypropiony1]-4-0-(6-amino- 6-deoxy-a-D-glucopyranosyl)-6-013-deoxy-4-C-methyl- 3-(methylamino)-ƒÀ-L-arabinopyranosyl]-2-deoxystreptamine
CHEMOTHERAPY APR. 1984
 VOL.32 S-3 CHEMOTHERAPY dihydro-4-oxo-7-(1-piperazinyl)-1, 8-naphthyridine- CHEMOTHERAPY APR. 1984 VOL.32 S-3 CHEMOTHERAPY Table 1 Implantation rates and post- implantation survival rates in females mated
VOL.32 S-3 CHEMOTHERAPY dihydro-4-oxo-7-(1-piperazinyl)-1, 8-naphthyridine- CHEMOTHERAPY APR. 1984 VOL.32 S-3 CHEMOTHERAPY Table 1 Implantation rates and post- implantation survival rates in females mated
 1.Streptococcus pneumoniae, Streptococcus pyogenes JC-1,S.aureus Smith,methicillin (DMPPC)- susceptible S. aureus subsp. aureus (MSSA) TR101, DMPPC-resistant S. aureus subsp. aureus (MRSA) TR102, Staphylococcus
1.Streptococcus pneumoniae, Streptococcus pyogenes JC-1,S.aureus Smith,methicillin (DMPPC)- susceptible S. aureus subsp. aureus (MSSA) TR101, DMPPC-resistant S. aureus subsp. aureus (MRSA) TR102, Staphylococcus
Key words:fatty acid,plant oil,staphylococcus aureus,skin care, atopic dermatitis
 Key words:fatty acid,plant oil,staphylococcus aureus,skin care, atopic dermatitis growth was monitored at 660 nm with a biophotorecorder. Table 1.Relative inhibitory effects of fatty acids,plant oils,
Key words:fatty acid,plant oil,staphylococcus aureus,skin care, atopic dermatitis growth was monitored at 660 nm with a biophotorecorder. Table 1.Relative inhibitory effects of fatty acids,plant oils,
CHEMOTHERAPY Proteus mirabilis GN-79 Escherichia coli No. 35 Proteus vulgaris GN-76 Pseudomonas aeruginosa No. 11 Escherichia coli ML-1410 RGN-823 Kle
 VOL. 29 NO.8 CHEMOTHERAPY 865 CHEMOTHERAPY Proteus mirabilis GN-79 Escherichia coli No. 35 Proteus vulgaris GN-76 Pseudomonas aeruginosa No. 11 Escherichia coli ML-1410 RGN-823 Klebsiella pneumoniae GN-69
VOL. 29 NO.8 CHEMOTHERAPY 865 CHEMOTHERAPY Proteus mirabilis GN-79 Escherichia coli No. 35 Proteus vulgaris GN-76 Pseudomonas aeruginosa No. 11 Escherichia coli ML-1410 RGN-823 Klebsiella pneumoniae GN-69
366 12 THE JAPANESE JOURNAL OF ANTIBIOTICS 65 6 Dec. 2012 1 8 DNA 2,3 16 12 20 171 2008 12 2010 11 2 3,558 4.44% 1.65% 1.17% 90% 9 Escherichia coli -
 Dec. 2012 THE JAPANESE JOURNAL OF ANTIBIOTICS 65 6 365 11 sita oxacin 1 1 1 1 1 1 2 2 3 3 1 1 1 2 3 2012 9 14 sita oxacin STFX 50 mg 10% 2008 1 2008 12 2010 11 2 STFX 1,452 91.4% 1,235/1,351 95.9% 466/486
Dec. 2012 THE JAPANESE JOURNAL OF ANTIBIOTICS 65 6 365 11 sita oxacin 1 1 1 1 1 1 2 2 3 3 1 1 1 2 3 2012 9 14 sita oxacin STFX 50 mg 10% 2008 1 2008 12 2010 11 2 STFX 1,452 91.4% 1,235/1,351 95.9% 466/486
Table 1. Antibacterial spectrum SBT ABPC ABPC CPZ : sulbactamiampicillin : ampicillin : cefoperazone
 Table 1. Antibacterial spectrum SBT ABPC ABPC CPZ : sulbactamiampicillin : ampicillin : cefoperazone (inoculum size= 106 CFU/ml) (Ĉ-lactamase producer : 2 strains) Fig. 1. Sensitivity distribution of
Table 1. Antibacterial spectrum SBT ABPC ABPC CPZ : sulbactamiampicillin : ampicillin : cefoperazone (inoculum size= 106 CFU/ml) (Ĉ-lactamase producer : 2 strains) Fig. 1. Sensitivity distribution of
Fig. 1 Chemical structure of KW-1070
 Fig. 1 Chemical structure of KW-1070 Fig. 2 Sensitivity distribution of clinical isolates Fig. 4 Sensitivity distribution of clinical isolates Fig. 3 Sensitivity distribution of clinical isolates Fig.
Fig. 1 Chemical structure of KW-1070 Fig. 2 Sensitivity distribution of clinical isolates Fig. 4 Sensitivity distribution of clinical isolates Fig. 3 Sensitivity distribution of clinical isolates Fig.
CHEMO THE RAPY OCT. 1994
 bilis (0.78), Proteus vulgaris (1.56), Enterococcus faecalis (3.13), Staphylococcus epidermidis (6.25), Klebsiella pneumoniae (6.25), Morganella morganii (6.25), Escherichia coli (12.5), Providencia rettgeri
bilis (0.78), Proteus vulgaris (1.56), Enterococcus faecalis (3.13), Staphylococcus epidermidis (6.25), Klebsiella pneumoniae (6.25), Morganella morganii (6.25), Escherichia coli (12.5), Providencia rettgeri
VOL.42 S-1
 CHEMOTHERAPY APR. 1994 VOL.42 S-1 CHEMOTHERAPY APR. 1994 Table 1. Criteria for evaluation of clinical efficacy by the Japanese Society of Oral and Maxillo-Facial Surgeons Grades of symptoms and numerical
CHEMOTHERAPY APR. 1994 VOL.42 S-1 CHEMOTHERAPY APR. 1994 Table 1. Criteria for evaluation of clinical efficacy by the Japanese Society of Oral and Maxillo-Facial Surgeons Grades of symptoms and numerical
400 46 THE JAPANESE JOURNAL OF ANTIBIOTICS 65 6 Dec. 2012 LVFX 100 mg 3 / 7 150 mg 2 / 7 2 2006 2008 9 LVFX PK PD 2009 7 100 mg 1 3 500 mg 1 1 AUC/MIC
 Dec. 2012 THE JAPANESE JOURNAL OF ANTIBIOTICS 65 6 399 45 2012 11 5 LVFX 500 mg 1 1 20 Chlamydia trachomatis C. trachomatismycoplasma genitalium M. genitalium LVFX 1 500 mg 1 1 7 22 22 C. trachomatis 17
Dec. 2012 THE JAPANESE JOURNAL OF ANTIBIOTICS 65 6 399 45 2012 11 5 LVFX 500 mg 1 1 20 Chlamydia trachomatis C. trachomatismycoplasma genitalium M. genitalium LVFX 1 500 mg 1 1 7 22 22 C. trachomatis 17
日本化学療法学会雑誌第64巻第4号
 β β Moraxella catarrhalisescherichia colicitrobacter Klebsiella pneumoniaeenterobacter cloacaeserratia marcescens Proteus Providencia Pseudomonas aeruginosaacinetobacter Bacteroides fragilis β β E. colik.
β β Moraxella catarrhalisescherichia colicitrobacter Klebsiella pneumoniaeenterobacter cloacaeserratia marcescens Proteus Providencia Pseudomonas aeruginosaacinetobacter Bacteroides fragilis β β E. colik.
CHEMOTHERAPY Fig. 1 Body weight changes of pregnant mice treated orally with AM- 715 Day of sestation
 CHEMOTHERAPY CHEMOTHERAPY Fig. 1 Body weight changes of pregnant mice treated orally with AM- 715 Day of sestation CHEMOTHERAPY Table 1 Preliminaly test of AM- 715 1): Mean } SD *: Significant difference
CHEMOTHERAPY CHEMOTHERAPY Fig. 1 Body weight changes of pregnant mice treated orally with AM- 715 Day of sestation CHEMOTHERAPY Table 1 Preliminaly test of AM- 715 1): Mean } SD *: Significant difference
 Key words: disinfectant, amphoteric surfactant, ethanol, Mycobacterium tuberculosis Table 1 Bactericidal effects of cresol soap and an amphoteric surfactant (ADG) against M. tuberculosis H37Rv : The numbers
Key words: disinfectant, amphoteric surfactant, ethanol, Mycobacterium tuberculosis Table 1 Bactericidal effects of cresol soap and an amphoteric surfactant (ADG) against M. tuberculosis H37Rv : The numbers
Oct THE JAPANESE JOURNAL OF ANTIBIOTICS Pseudomonas aeruginosa 186 P. aeruginosa piperacillin PIPC, taz
 Oct. 2016 THE JAPANESE JOURNAL OF ANTIBIOTICS 69 5 327 27 2013 2014 2016 7 5 2013 2014 Pseudomonas aeruginosa 186 P. aeruginosa piperacillin PIPC, tazobactam/piperacillin TAZ/PIPC, ceftazidime CAZ, cefepime
Oct. 2016 THE JAPANESE JOURNAL OF ANTIBIOTICS 69 5 327 27 2013 2014 2016 7 5 2013 2014 Pseudomonas aeruginosa 186 P. aeruginosa piperacillin PIPC, tazobactam/piperacillin TAZ/PIPC, ceftazidime CAZ, cefepime
Staphylococcus epidermidis Streptococcus pneumoniae Staphylococcus epidermidis Streptococcus pneumontae S. epidermidis Table 1. Summary of the organis
 Staphylococcus aureus S. aureus (MRSA) vancomycin (VCM), arbekacin (ABK) Streptococcus pneumoniae cefuzonam (CZON), cefpirome (CPR) S. pneumoniae Enterococcus faecalis ampicillin (ABPC), imipenem (IPM)
Staphylococcus aureus S. aureus (MRSA) vancomycin (VCM), arbekacin (ABK) Streptococcus pneumoniae cefuzonam (CZON), cefpirome (CPR) S. pneumoniae Enterococcus faecalis ampicillin (ABPC), imipenem (IPM)
CHEMOTHERAPY DEC Table 1 Antibacterial spectra of T-1982, CTT, CMZ, CTX, CPZ and CEZ 106 CFU/ml Note: P; Peptococcus, S; Streptococcus, G; Gaffk
 VOL. 30 S-3 CHEMOTHERAPY imeumoniae, Serratia marcescens, Proteus sp, CHEMOTHERAPY DEC. 1982 Table 1 Antibacterial spectra of T-1982, CTT, CMZ, CTX, CPZ and CEZ 106 CFU/ml Note: P; Peptococcus, S; Streptococcus,
VOL. 30 S-3 CHEMOTHERAPY imeumoniae, Serratia marcescens, Proteus sp, CHEMOTHERAPY DEC. 1982 Table 1 Antibacterial spectra of T-1982, CTT, CMZ, CTX, CPZ and CEZ 106 CFU/ml Note: P; Peptococcus, S; Streptococcus,
Key words: E. coli O 157: H7, fosfomycin, verotoxin, mouse infection
 Key words: E. coli O 157: H7, fosfomycin, verotoxin, mouse infection Table 1. Bacterial cell counts in feces of mice infected with Esclwrichia coli O 157: H7 NK2 before and during oral dosing with fosfomycin
Key words: E. coli O 157: H7, fosfomycin, verotoxin, mouse infection Table 1. Bacterial cell counts in feces of mice infected with Esclwrichia coli O 157: H7 NK2 before and during oral dosing with fosfomycin
VOL. 23 NO. 3 CHEMOTHERAPY 1067 Table 2 Sensitivity of gram positive cocci isolated from various diagnostic materials Table 3 Sensitivity of gram nega
 1066 CHEMOTHERAPY MAR. 1975 Table 1 Sensitivity of standard strains VOL. 23 NO. 3 CHEMOTHERAPY 1067 Table 2 Sensitivity of gram positive cocci isolated from various diagnostic materials Table 3 Sensitivity
1066 CHEMOTHERAPY MAR. 1975 Table 1 Sensitivity of standard strains VOL. 23 NO. 3 CHEMOTHERAPY 1067 Table 2 Sensitivity of gram positive cocci isolated from various diagnostic materials Table 3 Sensitivity
 Table 1.Quality control of MICs for reference strains Table 2.Antimicrobial activity of gatifloxacin against aerobic bacteria Table 4.Antimicrobial activity of gatifloxacin and other quinolones against
Table 1.Quality control of MICs for reference strains Table 2.Antimicrobial activity of gatifloxacin against aerobic bacteria Table 4.Antimicrobial activity of gatifloxacin and other quinolones against
Fig.2. Sensitivity distribution of clinical isolates of S. epidermidis (24 strains, 106 CFU/ml) Staphylococcus aureus Staphylococcus epider- midis Ent
 Fig.2. Sensitivity distribution of clinical isolates of S. epidermidis (24 strains, 106 CFU/ml) Staphylococcus aureus Staphylococcus epider- midis Enterococcus faecalis Klebsiella pneumoniae, Morganella
Fig.2. Sensitivity distribution of clinical isolates of S. epidermidis (24 strains, 106 CFU/ml) Staphylococcus aureus Staphylococcus epider- midis Enterococcus faecalis Klebsiella pneumoniae, Morganella
VOL. 40 S- 1 Table 1. Susceptibility of methicillin-resistant Staphylococcus aureus to meropenem Table 2. Coagulase typing of methicillin-resistant St
 CHEMOTHERAPY VOL. 40 S- 1 Table 1. Susceptibility of methicillin-resistant Staphylococcus aureus to meropenem Table 2. Coagulase typing of methicillin-resistant Staphylococcus aureus CHEMOTHERAPY Table
CHEMOTHERAPY VOL. 40 S- 1 Table 1. Susceptibility of methicillin-resistant Staphylococcus aureus to meropenem Table 2. Coagulase typing of methicillin-resistant Staphylococcus aureus CHEMOTHERAPY Table
CHEMOTHERAPY JUNE 1993 Table 1. Background of patients in pharmacokinetic study
 CHEMOTHERAPY JUNE 1993 Table 1. Background of patients in pharmacokinetic study VOL. 41 S 1 Table 2. Levels (Đg/ml or Đg/g) of S-1006 in serum, bile, and tissue (gallbladder) after oral administration
CHEMOTHERAPY JUNE 1993 Table 1. Background of patients in pharmacokinetic study VOL. 41 S 1 Table 2. Levels (Đg/ml or Đg/g) of S-1006 in serum, bile, and tissue (gallbladder) after oral administration
THE JAPANESE JOURNAL OF ANTIBIOTICS 63 13 243 ( 37 ) 2007 12 2008 5 19 863 methicillin-susceptible Staphylococcus aureus (MSSA) Escherichia coli levof
 242 ( 36 ) THE JAPANESE JOURNAL OF ANTIBIOTICS 63 _ 3 prulifloxacin * ** ** CMC * ** 2010 2 22 Prulifloxacin ulifloxacin (UFX) 3 1 2003 12 2004 5 19 534 2 2005 12 2006 5 19 805 3 THE JAPANESE JOURNAL OF
242 ( 36 ) THE JAPANESE JOURNAL OF ANTIBIOTICS 63 _ 3 prulifloxacin * ** ** CMC * ** 2010 2 22 Prulifloxacin ulifloxacin (UFX) 3 1 2003 12 2004 5 19 534 2 2005 12 2006 5 19 805 3 THE JAPANESE JOURNAL OF
 Table 1 Sensitivity distribution of clinical isolates 1. Escherichia coli Inoculum size: 106cells/ml 2. Klebsiella pneumoniae 3. Enterobacter cloacae 4. Serratia marcescens Inoculum size: 106cells/nil
Table 1 Sensitivity distribution of clinical isolates 1. Escherichia coli Inoculum size: 106cells/ml 2. Klebsiella pneumoniae 3. Enterobacter cloacae 4. Serratia marcescens Inoculum size: 106cells/nil
 Fig.1 MICs of penicillins against 24 strains of B. pertussis Fig.2 MICs of cepherns against 24 strains of B. pertussis Fig.3 MICs of macrolides against 24 strains of B. pertussis Fig.4 MICs of nalidixic
Fig.1 MICs of penicillins against 24 strains of B. pertussis Fig.2 MICs of cepherns against 24 strains of B. pertussis Fig.3 MICs of macrolides against 24 strains of B. pertussis Fig.4 MICs of nalidixic
CHEMOTHERAPY
 CHEMOTHERAPY CHEMOTHERAPY Table 1 Antibacterial activity of BRL 28500 against standard strains of bacteria Fig, 1 Sensitivity distribution of ABPC-resistant E. coli isolated from urinary tract Fig. 2 Sensitivity
CHEMOTHERAPY CHEMOTHERAPY Table 1 Antibacterial activity of BRL 28500 against standard strains of bacteria Fig, 1 Sensitivity distribution of ABPC-resistant E. coli isolated from urinary tract Fig. 2 Sensitivity
VOL. 34 S-2 CHEMOTH8RAPY 913
 VOL. 34 S-2 CHEMOTH8RAPY 913 914 CHEMOTHERAPY APR. 1986 Fig. 1 Chemical structure of T-2588 and T-2525 T- 2588 pivaloyloxymethyl (+ )- (6 R, 7 R)-7-[(Z)-2- (2-amino- 4-thiazolyl)-2-methox yiminoacetamido]-3-[(
VOL. 34 S-2 CHEMOTH8RAPY 913 914 CHEMOTHERAPY APR. 1986 Fig. 1 Chemical structure of T-2588 and T-2525 T- 2588 pivaloyloxymethyl (+ )- (6 R, 7 R)-7-[(Z)-2- (2-amino- 4-thiazolyl)-2-methox yiminoacetamido]-3-[(
Key Words: Klebsiella pneumoniae, CEP-AIS, MIC, "MBC", MIC of drugs in combination
 Key Words: Klebsiella pneumoniae, CEP-AIS, MIC, "MBC", MIC of drugs in combination Fig. 1. The following three kinds of overnight broth cultures of 10 strains of Kl. pneumoniae were inoculated on the agar
Key Words: Klebsiella pneumoniae, CEP-AIS, MIC, "MBC", MIC of drugs in combination Fig. 1. The following three kinds of overnight broth cultures of 10 strains of Kl. pneumoniae were inoculated on the agar
CHEMOTHERAPY Table 1 Clinical effect of Sultamicillin
 CHEMOTHERAPY CHEMOTHERAPY Table 1 Clinical effect of Sultamicillin CHEMOTHERAPY Fig. 1 MICs of sultamicillin against respiratory pathogenic Branhamella catarrhalis 62 strains, inoculum size 106CFU/m1 Fig.
CHEMOTHERAPY CHEMOTHERAPY Table 1 Clinical effect of Sultamicillin CHEMOTHERAPY Fig. 1 MICs of sultamicillin against respiratory pathogenic Branhamella catarrhalis 62 strains, inoculum size 106CFU/m1 Fig.
08-g-”O−}„j‹ê-4.02
 200( 96 ) THE JAPANESE JOURNAL OF ANTIBIOTICS 58 2 Apr. 585 1 17 MIC 1. (Transtracheal aspiration: TTA) TTA TTA TTA 1400 TTA (diffuse panbronchiolitis, DPB) 1) erythromycin(em) EM 12.5% 87.5% 2,3) EM EM
200( 96 ) THE JAPANESE JOURNAL OF ANTIBIOTICS 58 2 Apr. 585 1 17 MIC 1. (Transtracheal aspiration: TTA) TTA TTA TTA 1400 TTA (diffuse panbronchiolitis, DPB) 1) erythromycin(em) EM 12.5% 87.5% 2,3) EM EM
 Table 1. Antibacterial activity of cefdinir, cefixime, cefteram, cefuroxime, cefaclor and amoxicillin against standard strains Inoculum size: 108 cells/ml CFDN: cefdinir, CFIX: cefixime, CFTM: cefteram,
Table 1. Antibacterial activity of cefdinir, cefixime, cefteram, cefuroxime, cefaclor and amoxicillin against standard strains Inoculum size: 108 cells/ml CFDN: cefdinir, CFIX: cefixime, CFTM: cefteram,
Table 1. Antimicrobial drugs using for MIC
 Table 1. Antimicrobial drugs using for MIC Table 2. Susceptibilities determined with the VITEK 2 system and agar dilution reference by interpretive eategory for Staphylococcus aureus Table 3. Interpretive
Table 1. Antimicrobial drugs using for MIC Table 2. Susceptibilities determined with the VITEK 2 system and agar dilution reference by interpretive eategory for Staphylococcus aureus Table 3. Interpretive
 Table 1. Influence of urine ph on MBCs of new quinolones against Escherichia coli NIHJ JC-2 and Pseudomonas aeruginosa 18S; MBCs in urine were compared with those in Miieller-Hinton broth. Table 2. Influence
Table 1. Influence of urine ph on MBCs of new quinolones against Escherichia coli NIHJ JC-2 and Pseudomonas aeruginosa 18S; MBCs in urine were compared with those in Miieller-Hinton broth. Table 2. Influence
 CHEMOTHERAPY APRIL 1992 Table 2. Concentration of meropenem in human prostatic fluid Table 1. Background of 21 chronic complicated UTI cases * NB + BPH, NB + Kidney tumor, NB + Kidney tuberculosis Table
CHEMOTHERAPY APRIL 1992 Table 2. Concentration of meropenem in human prostatic fluid Table 1. Background of 21 chronic complicated UTI cases * NB + BPH, NB + Kidney tumor, NB + Kidney tuberculosis Table
CHEMOTHERAPY NOV Fig.1 Combined effect of drug A and drug B Fig. 2 MICs of CEPs (LMOX, CZX, CMX, CPZ) against 27 strains (inoculum size 106 CFU/
 CHEMOTHERAPY NOV. 1987 Fig.1 Combined effect of drug A and drug B Fig. 2 MICs of CEPs (LMOX, CZX, CMX, CPZ) against 27 strains (inoculum size 106 CFU/ml) of S. marcescens + Visible growth No visible growth
CHEMOTHERAPY NOV. 1987 Fig.1 Combined effect of drug A and drug B Fig. 2 MICs of CEPs (LMOX, CZX, CMX, CPZ) against 27 strains (inoculum size 106 CFU/ml) of S. marcescens + Visible growth No visible growth
CHEMOTHERAPY Table 1 Urinary excretion of mezlocillin Fig. 4 Urinary excretion of mezlocillin Fig. 3 Blood levels of mezlocillin
 CHEMOTHERAPY Fig. 2 Urinary excretion of mezlocillin Fig. 1 Blood levels of mezlocillin CHEMOTHERAPY Table 1 Urinary excretion of mezlocillin Fig. 4 Urinary excretion of mezlocillin Fig. 3 Blood levels
CHEMOTHERAPY Fig. 2 Urinary excretion of mezlocillin Fig. 1 Blood levels of mezlocillin CHEMOTHERAPY Table 1 Urinary excretion of mezlocillin Fig. 4 Urinary excretion of mezlocillin Fig. 3 Blood levels
VOL.48 NO.7 lase negative staphylococci, Escherichia coli, Klebsiella spp., Citrobacter freundii, Enterobacter spp., indole-positive Proteus, Serratia
 ceftazidime, cefpirome, cefepime, cefoperazone/sulbactam (2: 1), imipenem Staphylococcus aureus oxacillin coagulase negative staphylococci Escherichia coli piperacillin Klebsiellac spp. Citrobacter Pseudomonas
ceftazidime, cefpirome, cefepime, cefoperazone/sulbactam (2: 1), imipenem Staphylococcus aureus oxacillin coagulase negative staphylococci Escherichia coli piperacillin Klebsiellac spp. Citrobacter Pseudomonas
日本化学療法学会雑誌第61巻第6号
 β Moraxella catarrhalis Escherichia coli Citrobacter Klebsiella pneumoniae Enterobacter cloacae Serratia marcescens Proteus Pseudomonas aeruginosa Acinetobacter Bacteroides fragilis β Haemophilus influenzae
β Moraxella catarrhalis Escherichia coli Citrobacter Klebsiella pneumoniae Enterobacter cloacae Serratia marcescens Proteus Pseudomonas aeruginosa Acinetobacter Bacteroides fragilis β Haemophilus influenzae
Fig. 1 Chemical structure of TE-031 Code number: TE-031 Chemical name: (-) (3R, 4S, 5S, 6R, 7R, 9R, 11R, 12R, 13S, 14R)-4-[(2, 6-dideoxy-3-C-methyl-3-
 Fig. 1 Chemical structure of TE-031 Code number: TE-031 Chemical name: (-) (3R, 4S, 5S, 6R, 7R, 9R, 11R, 12R, 13S, 14R)-4-[(2, 6-dideoxy-3-C-methyl-3-O-methyl-a-L-ribo-hexopyranosyl) oxy]-14-ethyl-12,
Fig. 1 Chemical structure of TE-031 Code number: TE-031 Chemical name: (-) (3R, 4S, 5S, 6R, 7R, 9R, 11R, 12R, 13S, 14R)-4-[(2, 6-dideoxy-3-C-methyl-3-O-methyl-a-L-ribo-hexopyranosyl) oxy]-14-ethyl-12,
 VOL. 17 NO. 7 CHEMOTHERAPY 1305 1) W. BRumFirr et al. : Clinical and laboratory studies with carbenicillin. Lancet 1: 1289~ 1293, 1967 2) E. T. KNUDSEN et al. : A new semisynthetic penicillin active against
VOL. 17 NO. 7 CHEMOTHERAPY 1305 1) W. BRumFirr et al. : Clinical and laboratory studies with carbenicillin. Lancet 1: 1289~ 1293, 1967 2) E. T. KNUDSEN et al. : A new semisynthetic penicillin active against
VOL. 23 NO. 3 CHEMOTHERAPY 1379 Table 1 Susceptibility of clinical isolated strains to Tobramycin
 VOL. 23 NO. 3 CHEMOTHERAPY 1379 Table 1 Susceptibility of clinical isolated strains to Tobramycin 1380 CHEMOTHERAPY MAR. 1975 Table 2 Susceptibility of isolated Pseudomonas aeruginosa to various antibiotics
VOL. 23 NO. 3 CHEMOTHERAPY 1379 Table 1 Susceptibility of clinical isolated strains to Tobramycin 1380 CHEMOTHERAPY MAR. 1975 Table 2 Susceptibility of isolated Pseudomonas aeruginosa to various antibiotics
VOL.27 S-3 CHEMO THERAPY mido)-3-[[[1- (2-dimethylaminoethyl)-1H-tetrazol-5-yl] -thio] methyl]-ceph-3-em-4-carboxylic acid dihydro- S. aureus 209-P JC
![VOL.27 S-3 CHEMO THERAPY mido)-3-[[[1- (2-dimethylaminoethyl)-1H-tetrazol-5-yl] -thio] methyl]-ceph-3-em-4-carboxylic acid dihydro- S. aureus 209-P JC VOL.27 S-3 CHEMO THERAPY mido)-3-[[[1- (2-dimethylaminoethyl)-1H-tetrazol-5-yl] -thio] methyl]-ceph-3-em-4-carboxylic acid dihydro- S. aureus 209-P JC](/thumbs/95/124246112.jpg) VOL.27 S-3 CHEMO THERAPY mido)-3-[[[1- (2-dimethylaminoethyl)-1H-tetrazol-5-yl] -thio] methyl]-ceph-3-em-4-carboxylic acid dihydro- S. aureus 209-P JC, E. coli NIHJ JC-2 pneumoniae E. coli 106, K. pneumoniae
VOL.27 S-3 CHEMO THERAPY mido)-3-[[[1- (2-dimethylaminoethyl)-1H-tetrazol-5-yl] -thio] methyl]-ceph-3-em-4-carboxylic acid dihydro- S. aureus 209-P JC, E. coli NIHJ JC-2 pneumoniae E. coli 106, K. pneumoniae
03-b-„FŒ{›xŒ¾-4.02
 518( 30 ) THE JAPANESE JOURNAL OF ANTIBIOTICS 58 6 Dec. 25 2003 1. * * Dec. THE JAPANESE JOURNAL OF ANTIBIOTICS 58 6 519( 31 ) 9 5 2003 8 2004 7 14 565 701 258 (36.8%) 443 (63.2%) Staphylococcus aureus
518( 30 ) THE JAPANESE JOURNAL OF ANTIBIOTICS 58 6 Dec. 25 2003 1. * * Dec. THE JAPANESE JOURNAL OF ANTIBIOTICS 58 6 519( 31 ) 9 5 2003 8 2004 7 14 565 701 258 (36.8%) 443 (63.2%) Staphylococcus aureus
VOL. 43 NO. 4
 VOL. 43 NO. 4 Fig. 1. Frequency of Enterococcus species from complicated UTI, 1988-1992. the number * of Enterococcus species/the number of cases with complicated UTI. Fig. 3 Epidemiologic characteristics
VOL. 43 NO. 4 Fig. 1. Frequency of Enterococcus species from complicated UTI, 1988-1992. the number * of Enterococcus species/the number of cases with complicated UTI. Fig. 3 Epidemiologic characteristics
Table 1. St-VAc blockcopolymers Table 2. Stability of dispersion of blockcopolymers in unsaturated polyester
 97 Table 1. St-VAc blockcopolymers Table 2. Stability of dispersion of blockcopolymers in unsaturated polyester (a) (b) (c) Fig. 1. Phase reversions of poly (St-b-VAc) in unsaturated polyester. Fig. 2.
97 Table 1. St-VAc blockcopolymers Table 2. Stability of dispersion of blockcopolymers in unsaturated polyester (a) (b) (c) Fig. 1. Phase reversions of poly (St-b-VAc) in unsaturated polyester. Fig. 2.
Jpn.J.Leprosy 67, (1998)
 Jpn.J.Leprosy 67,287-291(1998) Fig.1. Macroscopic appearance of the cultured slides stained by the Ziehl-Neelsen method. Up to bottom: kept at 4 C, 18 weeks, cultured for 6 weeks, 8 weeks, 10 weeks, 12
Jpn.J.Leprosy 67,287-291(1998) Fig.1. Macroscopic appearance of the cultured slides stained by the Ziehl-Neelsen method. Up to bottom: kept at 4 C, 18 weeks, cultured for 6 weeks, 8 weeks, 10 weeks, 12
 CHEMOTHERAPY AUG. 1982 VOL. 30 NO. 8 CHEMOTHERAPY Fig.1 Relation between various-closis of cefazolin and detection rate of organisms in heart blood of dying mice with E. coli and P. aeruginosa infection
CHEMOTHERAPY AUG. 1982 VOL. 30 NO. 8 CHEMOTHERAPY Fig.1 Relation between various-closis of cefazolin and detection rate of organisms in heart blood of dying mice with E. coli and P. aeruginosa infection
THE JAPANESE JOURNAL OF ANTIBIOTICS 48-8 Enterococcus avium 5Š, Corynebacterium xerosis 10Š, Corynebacterium pseudodiphtheriticum 10Š, Corynebacterium
 THE JAPANESE JOURNAL OF ANTIBIOTICS 48-8 Enterobacter spp., Serratia spp., Burkholderia cepacia, Flavobacterium spp., Alcaligenes spp. THE JAPANESE JOURNAL OF ANTIBIOTICS 48-8 Enterococcus avium 5Š, Corynebacterium
THE JAPANESE JOURNAL OF ANTIBIOTICS 48-8 Enterobacter spp., Serratia spp., Burkholderia cepacia, Flavobacterium spp., Alcaligenes spp. THE JAPANESE JOURNAL OF ANTIBIOTICS 48-8 Enterococcus avium 5Š, Corynebacterium
Key words: change serotype, Pseudomonas aeruginosa anti-pseudomonal drug,
 Key words: change serotype, Pseudomonas aeruginosa anti-pseudomonal drug, Table 1 Incidence of changes in serotype of Pseudomonas aeruginosa isolates induced by anti-pseudomonal drugs Number of isolates
Key words: change serotype, Pseudomonas aeruginosa anti-pseudomonal drug, Table 1 Incidence of changes in serotype of Pseudomonas aeruginosa isolates induced by anti-pseudomonal drugs Number of isolates
THE JAPANESE JOURNAL OF ANTIBIOTICS 67 3 June 2014 STFX mg mg mg mg mg 1
 June 2014 THE JAPANESE JOURNAL OF ANTIBIOTICS 67 3 175 29 1 2 2 2 2 2 3 4 4 5 5 2 2 1 2 3 4 5 2014 4 15 STFX 50 mg 10% 2011 8 1 100 mg 1 1 2011 12 2013 5 226 1,186 1,089 1,069 2.11% 23/1,089 1.10% 12/1,089
June 2014 THE JAPANESE JOURNAL OF ANTIBIOTICS 67 3 175 29 1 2 2 2 2 2 3 4 4 5 5 2 2 1 2 3 4 5 2014 4 15 STFX 50 mg 10% 2011 8 1 100 mg 1 1 2011 12 2013 5 226 1,186 1,089 1,069 2.11% 23/1,089 1.10% 12/1,089
2108 CHEMOTHERAPY SEPT Table 1 Antimicrobial spectrum Fig. 1
 2108 CHEMOTHERAPY SEPT. 1977 Table 1 Antimicrobial spectrum Fig. 1 VOL. 25 NO. 7 CHEM 014 HERAPY 2109 Table 2 Susceptibility distribution of Staphylococcus aureus to aminoglycosides (54 strains) Table
2108 CHEMOTHERAPY SEPT. 1977 Table 1 Antimicrobial spectrum Fig. 1 VOL. 25 NO. 7 CHEM 014 HERAPY 2109 Table 2 Susceptibility distribution of Staphylococcus aureus to aminoglycosides (54 strains) Table
Experimental and Clinical Studies of Pregnant Hypertension Takashi SHIMAZU Department of Obstetrics and Gynecology, Osaka City University Medical Scho
 Experimental and Clinical Studies of Pregnant Hypertension Takashi SHIMAZU Department of Obstetrics and Gynecology, Osaka City University Medical School While the problem of late pregnant hypertension
Experimental and Clinical Studies of Pregnant Hypertension Takashi SHIMAZU Department of Obstetrics and Gynecology, Osaka City University Medical School While the problem of late pregnant hypertension
Key words : 7432-S, Oral cephem, Urinary tract infection Fig. 1. Chemical structure of 7432-S.
 Key words : 7432-S, Oral cephem, Urinary tract infection Fig. 1. Chemical structure of 7432-S. Table 1. Clinical summary of acute uncomplicated cystitis patients treated with 7432-S UTI : Criteria by the
Key words : 7432-S, Oral cephem, Urinary tract infection Fig. 1. Chemical structure of 7432-S. Table 1. Clinical summary of acute uncomplicated cystitis patients treated with 7432-S UTI : Criteria by the
CHEMOTHERAPY FEB Table 1 Background of volunteers
 CHEMOTHERAPY FEB. 1985 Table 1 Background of volunteers Table 3 Reproducibility of saisomicln In the EIA and the RIA Fig.1 Comparison of the EIA with the RIA or bioassay of sisomicin Table 4 Blood levels
CHEMOTHERAPY FEB. 1985 Table 1 Background of volunteers Table 3 Reproducibility of saisomicln In the EIA and the RIA Fig.1 Comparison of the EIA with the RIA or bioassay of sisomicin Table 4 Blood levels
 Helicobacter pylori Helicobacter pylori Helkobacter pylori erythromycin (EM) Mueller Hinton broth (Difco) H. pylori Staphylococcus aureus ATCC 29213 Escherichia coli ATCC25922 amoxicillin (AMPC), clarithromycin
Helicobacter pylori Helicobacter pylori Helkobacter pylori erythromycin (EM) Mueller Hinton broth (Difco) H. pylori Staphylococcus aureus ATCC 29213 Escherichia coli ATCC25922 amoxicillin (AMPC), clarithromycin
VOL.27S-1 CHEMOTHERAPY 109 Klebsiella, Proteus, Pseudomonas Streptococcus Fig. 1 Concentration in blood and in CSF after intravenous drip infusion of
 VOL.27S-1 CHEMOTHERAPY 109 Klebsiella, Proteus, Pseudomonas Streptococcus Fig. 1 Concentration in blood and in CSF after intravenous drip infusion of 4.9 g Mezlocillin Streptococcus pneumonzae pneumoniae
VOL.27S-1 CHEMOTHERAPY 109 Klebsiella, Proteus, Pseudomonas Streptococcus Fig. 1 Concentration in blood and in CSF after intravenous drip infusion of 4.9 g Mezlocillin Streptococcus pneumonzae pneumoniae
CHEMOTHERAPY JUNE 1986
 VOL. 34 S-3 CHEMOTHERAPY Fig. 1 Structural formula of L-105 CHEMOTHERAPY JUNE 1986 VOL. 34 S-3 CHEMOTHERAPY Table 1 Antibacterial spectra of L-105 against gram negative anaerobic rods Inoculum 106 cells/ml
VOL. 34 S-3 CHEMOTHERAPY Fig. 1 Structural formula of L-105 CHEMOTHERAPY JUNE 1986 VOL. 34 S-3 CHEMOTHERAPY Table 1 Antibacterial spectra of L-105 against gram negative anaerobic rods Inoculum 106 cells/ml
 Key words : R-plasmid, Urinary tract infection, E. coli Fig. 1. MIC distribution against E. coli isolated from urinary tract (366 strains) and isolation - frequencies of drug-resistant strains Table 1.
Key words : R-plasmid, Urinary tract infection, E. coli Fig. 1. MIC distribution against E. coli isolated from urinary tract (366 strains) and isolation - frequencies of drug-resistant strains Table 1.
Table1MIC of BAY o 9867 against standard strains
 Table1MIC of BAY o 9867 against standard strains Fig.2Cumulative and Distribution Curves of MIC (S.aureus 54 strains) 106cfu/ml Fig.3Correlogram of MIC (S.aureus 54 strains) CHEMOTHERAPY 451 Fig.4Cumulative
Table1MIC of BAY o 9867 against standard strains Fig.2Cumulative and Distribution Curves of MIC (S.aureus 54 strains) 106cfu/ml Fig.3Correlogram of MIC (S.aureus 54 strains) CHEMOTHERAPY 451 Fig.4Cumulative
特殊病態下感染症における抗菌薬治験の手引き作成委員会報告書(案)
 VOL.51 NO.6 JUNE 2003 JUNE 2003 9) Niederman MS et al: Guidelines for the management of adults with community-acquired pneumonia. Diagnosis, assessment of severity, antimicrobial therapy and prevention.
VOL.51 NO.6 JUNE 2003 JUNE 2003 9) Niederman MS et al: Guidelines for the management of adults with community-acquired pneumonia. Diagnosis, assessment of severity, antimicrobial therapy and prevention.
CHEMOTHERAPY MAY. 1988
 CHEMOTHERAPY MAY. 1988 CHEMOTHERAPY Fig. 1 Chemical structure CHEMOTHERAPY MAY. 1988 VOL.36 5-1 CHEMOTHERAPY CHEMOTHERAPY MAY. 1988 VOL.36 S-1 CHEMOTHERAPY CHEMOTHERAPY MAY. 1988 VOL.36 S-1 CHEMOTHERAPY
CHEMOTHERAPY MAY. 1988 CHEMOTHERAPY Fig. 1 Chemical structure CHEMOTHERAPY MAY. 1988 VOL.36 5-1 CHEMOTHERAPY CHEMOTHERAPY MAY. 1988 VOL.36 S-1 CHEMOTHERAPY CHEMOTHERAPY MAY. 1988 VOL.36 S-1 CHEMOTHERAPY
Fig. 1 Chemical structure of norfioxacin (AM-715)
 Fig. 1 Chemical structure of norfioxacin (AM-715) Table 1 Serum and biliary concentration of norfloxacin (AM-715) Table 2 Protocol for clinical evaluation of norfloxacin (AM-715) in the treatment of biliary
Fig. 1 Chemical structure of norfioxacin (AM-715) Table 1 Serum and biliary concentration of norfloxacin (AM-715) Table 2 Protocol for clinical evaluation of norfloxacin (AM-715) in the treatment of biliary
 CHEMOTHERAPY 218 5. NFLXの 血 清お よび胆 汁中 濃度の 測定 NFLXの50mg/kgを1回 後,経 coli NIHJ JC-2を 定 した な お,血 た 度 をE. 検 定 菌 とす る 薄 層 デ ィ ス ク法 で 測 (2) NFLX治 E. 療群 coli接 種6時 50mg/kg経 間 後 の 家 兎 にNFLXを25お 液 は 耳 静 脈 よ り,胆 村
CHEMOTHERAPY 218 5. NFLXの 血 清お よび胆 汁中 濃度の 測定 NFLXの50mg/kgを1回 後,経 coli NIHJ JC-2を 定 した な お,血 た 度 をE. 検 定 菌 とす る 薄 層 デ ィ ス ク法 で 測 (2) NFLX治 E. 療群 coli接 種6時 50mg/kg経 間 後 の 家 兎 にNFLXを25お 液 は 耳 静 脈 よ り,胆 村
