Microsoft Word docx
|
|
|
- しのぶ いさやま
- 7 years ago
- Views:
Transcription
1 m m kg t s I A T K n mol I v cd SISI SI 10 1 SI molecular weight1 12 C 112 relative molecular mass of a substance g 1 mol dalton 12 C 112 SI x10-27 kg Da SI SI SI 10-1 deci d 10 deca da 10-2 centi c 10 2 hecto h 10-3 milli m 10 3 kilo k 10-6 micro µ 10 6 mega M 10-9 nano n 10 9 giga G pico p tera T femto f peta P atto a exa E
2 ph deflectional methodnull method precision accuracy
3 µ σ ± σ 68%± 2σ± 3σ 95%99.7% n x I i = 1n σ s s > σ 1.1.2n n 1 n s σ 100s RSDCV s 2 = V n µ µ σ
4 95% P = 0.05 µ ± σ s µφ = n 1t t µ %P = 0.05 t φ t φ t y abc y = f a, b, c,δy yab s y s a s b y = kab yab s y ys a as b b 4
5 1.1.8 y = ka n : F F o V 1 > V F o F 95% P = 0.05 F %P = 0.05 F φ 1 φ
6 F 0 F 0 F 95% F 0 > F 95% F t µ µ µ t t o t t o t 95%µ t o > t 95% t φφ = n t o t o t t φ φ = n 1 +n 2 2 t o t 1 95% t o > t 95% 6
7 Dixon x 1 x 2 Q o x min x max Q o Q 90%P = 0.10 Q Q o > Q 90% x 1 Q o Q %P = 0.10 Q n Q x y y x a b Y y x a b y y y a b
8 x i y i r r 1 < r < 1 r = 1 r = 1 r > r 1 r t o t t t φ = n 2 t o > t y x a b sa sb b ± tsbt : φ = n 2 y x y = f yy =ax+b y y y gnuplot y= hx 1 M.L.McGlashan J.C. Miller, J.N.Miller
9 AEx TD EIn TC 1-2-1d a b c d e f g h abcd efgh
10 a 1-2-1b 1-2-1b ml 10
11 e f ) 0.01 ml
12 h 1-2-1g P P µl
13 1-2-4 P P-2P-10 P-20P-100 P-200P-1000 P mm 23 mm 24 mm 36 mm P-2P
14 A B M Mg Mg Mg A-D Mg mg 0.1 mg 1 g 10 % 1 ± 0.1 g 0.1 mg g 14
15 W V W o W 1 W=W 1 W o W V 15
16 20 C 20 C 1-2-1a 1-2-1b c c V a g cm 3 t C b c 10 5 t C % Mw C V W V W ±10%0.1 mg c real C real C f C real 16
17 C real C f KIO C Pww% W d V s 17
18 1.2.5 P d P d C Mw dg ml 1 Pww % M HCl HNO H 2 SO H 3 PO CH 3 COOH NH HClO H 2 O ph ph H ph x ph RTF A B Nernst x s AgAgCl ph ph ph 18
19 1.2.7 T ph A ph ph 4 9 ph B ph HA HA H + + A K a
20 HA A C K a HA A logk a = pk a HA H 2 O H + + OH K w BOH BOH B + + OH HA BOH C A HA C B BOH A B )
21 HA C B = 0C A K a >> M 2 C A >> K a =OH HA αa = αc A H Ostwald C A << K a K a C A << M 2 OH C A = 10 6 M HCN K a = M1.2.19H + ph = ph 7 B H + B K a B H + B BOH C A = C B leveling effect K a HA C A C A BOH C A H + = C A OH = C B H + H 3 O + OH HCl ΔC B ph ΔpH logδc B C A > C B H + H 3 O + H 2 O H 2 O OH HA HA ΔHA= ΔA ΔC B ΔH + << ΔC B H ΔH + = ΔC B H 2 O ΔOH = ΔC B β 21
22 H HA A K a = H + C A H + H 3 O + H 2 O H 2 O OH ph pk a HA ph ph pk a ± 1 pk a 2 ph γ RTlnγ γ = 1 γ < 1 (r) Debye-HückelA M 1/2 25 Cz I i z i C i 0.1 M NaCl I 0.1 M0.1 M Na 2 SO 4 I
23 M I B M 1/2 m 1 NaCl = m Davies I γ K a < I pk a pk a I HA C HA MV HA ml A C A MV A ml NaH 2 PO 4 Na 2 HPO 4 ph 311 ph Henderson-Hasselbalch ph ph ph B C B MV B ml HA C HA MV HA ml 23
24 Na 2 HPO 4 + HCl CH 3 COONa + HClTris + HCl Tris H 2 NCCH 2 OH 3 B + H K a ph C B V B > C HA V HA ph ph ph HA C HA MV HA ml BOH (C B MV B ml) NaH 2 PO 4 + NaOH + NaOHNH 4 Cl + NaOHH 3 BO 3 + NaOH AH K a ph C HA V HA > C B V B ph ph ph + Na 2 HPO 4 MacIlvaine ph Na K + SO 4 2 NO 3 pk a 1-2-3Ca 2+ Fe 2+/ pk a pk a 2.15 H 3 PO HOOCCH 2 NH HOC(COOH)(CH 2 COOH) HOC(COOH)(CH 2 COOH)CH 2 COO CH 3 COOH 6.15 MES 6.40 HOC(COOH)(CH COO )
25 ph CA ph ph I pka ph [HA] [A ] NaCl ph ph H + C B pk a C A = 0.1 M ph ph logαc A C B = C A 2 phph eq H + OH pk a ph ph eq/ K a pk a < C A K a 25
26 ph eq2 = C A 2 pk a ph eq = OH ph C B = C A phph eq C A ph eq = pk W 2 = ph eq > K a >> [H + ] eq pk a HA 1 C A,1 K a,1 HA 2 C A,2 K a, pk a,1 << pk a,2 HA HA 1 26
27 C A,1 HA 2 pk a,1 pk a, H 2 A 2HA H 2 A A 2 H 2 A a b C A H 2 A BOH( C B ) a,b H 2 AHA A 2 C A K a,1 K a, H 2 A BOH (1.2.38) pk a,1 >> pk a,2 K a,1 H + >> K a,2 H K a,2 H C A,1 = C A,2 pk a,1 >> pk a,2 2HA H 2 A A 2 C A H 2 A HA ph ph In InH In ph
28 InH pk a ph ph ph pk a ph ph 7 ph 4 ph ph ph ph ph V ph
29 λ = nmλ = nm πnd I C M λ ελ M 1 cm 1 λaλ tλ Tλ Lambert-Beer Aλ C Aλλ λ max εε max ε max λ max π π* ε max > 2000-C=C-CO- ε max > 10000ε max > 100n π*-co-no 2 n ε max < 100 λ max ε max ΔE = hcλbohr 29
30 π n p ε max π π* n π*λ max σ π d* d σ*π* d d d ε max = mM = 1 cm ε max > λ(nm) log t C t F dCC
31 F F A F A = A = A t t = 0 t = W D 2 λ > 350 nm λ < 360 nm
32 NO nm Excel p TARE g 10 ml 32
33 W g 9 W 2 W 10 W W 0 90P = 0.10Dixon Dixon W W 0 n 10( )(s) c ml V ml 95%F 95% V 10.0 ml µl 200 µl 20 µl P-1000P-200P µl 1000 µl 1000 µl W g 9 W 2 W 0 3 W W 0 W W 0 90(P = 0.10) W W 0 n 10( )(s) coefficient of variation; 33
34 CV V ml% = 100V V o V o ; V o CV O µl CV P P P ph ph ph CLR + CAL ph F + CAL H 3 PO 4 ; Mw M f B 3L ph ph 4 MR 0.1 g 45 ml 50 ml25 PP 0.5 g 45 ml 50 ml25 34
35 !! 1.2 g 100 ml Fill-up P ml 10 ml 40 ml M NaOH M NaOH ml W mg 0.1 M NaOH f B H 3 PO 4 NaOH 0.1 M NaOH 1 ml = mg H 3 PO W wmg Pww% ph pk a,2 H 2 PO 4 pk a ph pk a,1 H 3 PO 4 pk a C A W (40 ml) ph pk a,3 HPO 4 2 pk a C A ΔpH ΔV B ΔV B ΔpH ph β ΔC B ΔpH ΔC B = C B f B ΔV B V t β V t ml 35
36 β C B f B ΔV B ΔpHV t ph ph ph pk a Excel ph etylenediamine tetraacetic acid, EDTA chelatometry EDTAH 4 Y H 4 Y H 3 Y H Y 2 HY 3 Y 4 Y 4 11 PtIV FeIIIY 4 Lewis H + Y 4 M n+ EDTA K MY,app K MY M n+ Y 4 α H Y t Y 4 EDTA K 1 pk 1 = 2.0K 2 pk 2 = 2.67K 3 pk 3 = 6.16K 4 pk 4 = α H ph K MY,app ph ph EBT
37 Mg 2+ EDTA logk MY 8.69 ph10 ph 11 Mg(OH) 2 Mg 2+ ph 1011 EBT Ca 2+ logk MY ph 13 ph NNMg 2+ Ca 2+ Mg 2+ Mg 2+ Ca 2+ ph 1213 Mg 2+ Ca 2+ EDTA ph H + Ca logα H ph Mg 2+ CaCO 3 Mw: ppm ppm = parts per million = M EDTA f E EDTA2Na2H 2 OMw g ml 6 Zn 25 6L 0.01 M f Mg MgCl 2 6H 2 OMw g mledta 25 EBT 0.1% 1:1 50mL 14 g 114 ml 200 ml; % 10 g ml25 10%NaOH 20 g ml25 NN K 2 SO ml 0.01 M 1.00 mlph 10 1 mlebt 5, M EDTA V E,t ml Mg 2+ EBT MgCl 2 37
38 ml 10% 1 ml10% 35 NN 0.05 g 0.01 M EDTA V E,Ca mlnn NN V E,t ml0.01 M EDTA f E 0.01 M f Mg EDTA CaCO M EDTA 1mL = mg CaCO 3 ppm= f E V E,t 1.00f Mg V E,Ca ml 0.01 M EDTA f E ppm = f E V E,Ca EDTA Mg 2+ Ca 2+ logk MY α H ph ph logk MY,app K MY,app C C L-ascorbic acid, AsAdehydroascorbate; DAsA AsA 2,6-2,6-dichlorophenolindophenol; DCIP 38
39 1.3.A1 AsA DAsA 1.3.A2 DCIP 2,6-dichloro-4-hydroxy-N-(4-hydroxyohenyl)-aniline DCIP DCIP AsA AsA DCIP 1.3.A3 DCIP AsA DCIP KIO 3 DCIP DCIP AsA IO IO 3 + 5e + 6H + 12 I 2 +3H 2 O 1.3.A4 6 6 IO 3 + 6e + 6H + I +3H 2 O 1.3.A5 3 M 4 IO 3 + 4e + Cl + 6H + ICl +3H 2 O AsA I 2 KIO 3 3AsA + KIO 3 3DAsA KI + 3H 2 O AsA 13KIO A6 1.3.A7 KI KIO 3 KI I 2 5KI + KIO 3 + 6H + 3I 2 + 6K + + 3H 2 O 1.3.A8 I 2 I 2 KI I 3 I 3 I 2 KIO 3 AsA 39
40 5% HPO 3 ; Mw %150g 2.85 L 25 2% 5% 40 ml 60 ml AsA 4mg100mL; 0.2 mm AsAC 6 H 8 O 6 ; Mw mg 2% 500 ml 100 ml AsA 6% KI KIMw g 100 ml 25 5 mm KIO 3 KIO 3 Mw g = W g 500 ml 5 mm KIO 3 W 25 f (KIO 3 ) = W mm KIO 3 5 mm KIO 3 20 ml 500 L 0.5% 0.5 g 100 ml mm DCIP DCIP-Na2H 2 OC 12 H 6 Cl 2 NNaO 2 2H 2 O Mw g 3 L ml AsA 20 ml 6% KI 1 ml mm KIO 3 VKIO 3 mlasa AsA C mgmlf KIO mm KIO 3 1 ml = = mg AsA 1.3.A8 C mgml = fKIO 3 VKIO A9 100 ml AsA 20 ml DCIP (DCIP 1mL AsA A [mgml]dcip V(DCIP) [ml] AmgmL = 20.0CVDCIP 1.3.A10 40
41 5% ml 5% 7.0 ml 8.0 ml d = ml V S mlv S DCIP ml AsA AsA 0.51 mg V S AsA mg100 ml AsA mg100 ml = AV S DCIP100dV S 1.3.A11 AsA DCIP AsA AsA I 2 AsA KIO 3 AsA sucrose glucose fructose (H 2 O 2 ) 41
42 glucose gluconic acid H 2 O 2 3,3,5,5 -(TMB) TMB π π* 0.20 mgml C 12 H 22 O 11 : Mw = g 500 ml 25 5 ml 0.1 mgml C 6 H 12 O 6 Mw = g 1000 ml 25 5 ml 0.1M ph M ph ml mg2000 U100 ml 0.1 M ph ml 45 mg 9 ml 0.1M ph TMB TMB2HClMw mg 9 ml 25 GPO 36 mg 90 ml 0.1M ph6.0 9 mltmb 9 ml 0.1M ph ml 25 42
43 20 ml 1 M H 2 SO ml H 2 SO 4 11 = 200 ml 1 M18 M)25 3 ml 0.1 mgml 0.2 mgml P O M H 2 SO ml 450 nm A r r > p.46 mg100 ml µ 43
44 glc suc O H 2 SO
45 Excel Office OK or AZ Q 0 = Max-MinMaxMin =data 2 data1max-min) data1data2 Max-Min or F4 Q Q 0 =AVERAGE=STDEV = AVERAGE STDEV = F 0 F = F F 0 F t t = t 0, t = t t 0 t t 0 t 0 45
46 R R r Memo 46
A9RF112.tmp.pdf
 9 1-1 9 9 10 11 13 17 1-2 18 18 19 20 21 21 22 23 24 26 2-1 26 26 26 30 33 35 2-2 36 36 38 40 44 44 45 3-1 45 45 47 49 51 53 58 3-2 59 59 60 62 64 68 69 70 4-1 70 70 72 4-2 73 73 74 74 75 76 77 77 79 80
9 1-1 9 9 10 11 13 17 1-2 18 18 19 20 21 21 22 23 24 26 2-1 26 26 26 30 33 35 2-2 36 36 38 40 44 44 45 3-1 45 45 47 49 51 53 58 3-2 59 59 60 62 64 68 69 70 4-1 70 70 72 4-2 73 73 74 74 75 76 77 77 79 80
H22応用物理化学演習1_濃度.ppt
 1 2 4/12 4/19 4/27 5/10 5/17 5/24 5/31 (20 ) (20 ) (10 ) (50 ) 3 (mole fraction) X = (mol) (mol) i n 1, n 2,, n x N i X i = n i = n i n 1 + n 2 + + n x N 4 (molarity, M) 1 dm 3 ( L) (mol) (mol/l) = 1 L
1 2 4/12 4/19 4/27 5/10 5/17 5/24 5/31 (20 ) (20 ) (10 ) (50 ) 3 (mole fraction) X = (mol) (mol) i n 1, n 2,, n x N i X i = n i = n i n 1 + n 2 + + n x N 4 (molarity, M) 1 dm 3 ( L) (mol) (mol/l) = 1 L
Microsoft Word - 目次注意事項2.doc
 I II 2009 1. 2. 12 50 3. 1. 2. 3 ( 0.1g, 0.01g, 0.001g ) 3. 4. 1. 2. ()A4 3. 000005 19 5 11 () 19 5 15 () EDTA " # $ 1000 500 =... mol/l Web 1) p.159 2) 1987pp.14-15 3) 1989pp.58-60 4) http://www.chem.zenkyo.h.kyoto-u.ac.jp/operation/
I II 2009 1. 2. 12 50 3. 1. 2. 3 ( 0.1g, 0.01g, 0.001g ) 3. 4. 1. 2. ()A4 3. 000005 19 5 11 () 19 5 15 () EDTA " # $ 1000 500 =... mol/l Web 1) p.159 2) 1987pp.14-15 3) 1989pp.58-60 4) http://www.chem.zenkyo.h.kyoto-u.ac.jp/operation/
2 Zn Zn + MnO 2 () 2 O 2 2 H2 O + O 2 O 2 MnO 2 2 KClO 3 2 KCl + 3 O 2 O 3 or 3 O 2 2 O 3 N 2 () NH 4 NO 2 2 O + N 2 ( ) MnO HCl Mn O + CaCl(ClO
 1 [1]. Zn + 2 H + Zn 2+,. K Ca Na Mg Al Zn Fe Ni Sn Pb H Cu Hg Ag Pt Au H (H + ),,. [2] ( ) ( ) CO 2, S, SO 2, NH 3 () + () () + () FeS Fe S ( ) + ( ) ( ) + ( ) 2 NH 4 Cl + Ca(OH) 2 Ca O + 2 NH 3,.,,.,,.,.
1 [1]. Zn + 2 H + Zn 2+,. K Ca Na Mg Al Zn Fe Ni Sn Pb H Cu Hg Ag Pt Au H (H + ),,. [2] ( ) ( ) CO 2, S, SO 2, NH 3 () + () () + () FeS Fe S ( ) + ( ) ( ) + ( ) 2 NH 4 Cl + Ca(OH) 2 Ca O + 2 NH 3,.,,.,,.,.
0 s T (s) /CR () v 2 /v v 2 v = T (jω) = + jωcr (2) = + (ωcr) 2 ω v R=Ω C=F (b) db db( ) v 2 20 log 0 [db] (3) v R v C v 2 (a) ω (b) : v o v o =
![0 s T (s) /CR () v 2 /v v 2 v = T (jω) = + jωcr (2) = + (ωcr) 2 ω v R=Ω C=F (b) db db( ) v 2 20 log 0 [db] (3) v R v C v 2 (a) ω (b) : v o v o = 0 s T (s) /CR () v 2 /v v 2 v = T (jω) = + jωcr (2) = + (ωcr) 2 ω v R=Ω C=F (b) db db( ) v 2 20 log 0 [db] (3) v R v C v 2 (a) ω (b) : v o v o =](/thumbs/91/105491855.jpg) RC LC RC 5 2 RC 2 2. /sc sl ( ) s = jω j j ω [rad/s] : C L R sc sl R 2.2 T (s) ( T (s) = = /CR ) + scr s + /CR () 0 s T (s) /CR () v 2 /v v 2 v = T (jω) = + jωcr (2) = + (ωcr) 2 ω v R=Ω C=F (b) db db(
RC LC RC 5 2 RC 2 2. /sc sl ( ) s = jω j j ω [rad/s] : C L R sc sl R 2.2 T (s) ( T (s) = = /CR ) + scr s + /CR () 0 s T (s) /CR () v 2 /v v 2 v = T (jω) = + jωcr (2) = + (ωcr) 2 ω v R=Ω C=F (b) db db(
物理化学I-第11回(13).ppt
 I- 11-1 10 10.5 H 3 O + (aq) + OH (aq) + Na + (aq) + Cl (aq) Na + (aq) + Cl (aq) + 2H 2 O(l) - or - [A] V A = [B] V B 22 mol dm 3 equiv dm 3 - H 2 SO 4 (aq) + 2NaOH(aq) 2Na + (aq) + SO 4 2 (aq) + 2H 2
I- 11-1 10 10.5 H 3 O + (aq) + OH (aq) + Na + (aq) + Cl (aq) Na + (aq) + Cl (aq) + 2H 2 O(l) - or - [A] V A = [B] V B 22 mol dm 3 equiv dm 3 - H 2 SO 4 (aq) + 2NaOH(aq) 2Na + (aq) + SO 4 2 (aq) + 2H 2
後期化学_04_酸塩基pH
 2011 ( ) ph H3O + H + H3O + HCl H3O + HCl + H2O H3O + + Cl HCl H + + Cl OH OH NaOH OH NaOH Na + + OH NH3 OH NH3 + H2O NH4 + + OH 1 H + OH H + H + * 1 NH3 HCl NH4Cl NH4Cl NH3 + Cl NH3 + HCl NH4 + + Cl.
2011 ( ) ph H3O + H + H3O + HCl H3O + HCl + H2O H3O + + Cl HCl H + + Cl OH OH NaOH OH NaOH Na + + OH NH3 OH NH3 + H2O NH4 + + OH 1 H + OH H + H + * 1 NH3 HCl NH4Cl NH4Cl NH3 + Cl NH3 + HCl NH4 + + Cl.
untitled
 (1) 100 100 60% (2) (3) - 1 - 1 2 3 4 100 200-2 - 1 2 3-3 - 4 5 6 7......... (1) (2) (3) 1) 2) 3) 8(5) - 4 - 0.5 27.3 3 0.05 27.30 4 0.005 Système International d'unités 7218 1 (1) Pas Pas J/molK J/(molK)
(1) 100 100 60% (2) (3) - 1 - 1 2 3 4 100 200-2 - 1 2 3-3 - 4 5 6 7......... (1) (2) (3) 1) 2) 3) 8(5) - 4 - 0.5 27.3 3 0.05 27.30 4 0.005 Système International d'unités 7218 1 (1) Pas Pas J/molK J/(molK)
3章 問題・略解
 S S W R S O( l) O( ) c Jg g J Jg S R J 7. K.9 JK S W S R S JK S S R J 7. K.9JK 4 (a) -Tice 7.K T ice T N 77 K S R.9 JK 4. JK T T ice N.6JK S W S R S JK S S.6JK R (b) S R JK S.6 JK T T ice N 6 O( c) O(
S S W R S O( l) O( ) c Jg g J Jg S R J 7. K.9 JK S W S R S JK S S R J 7. K.9JK 4 (a) -Tice 7.K T ice T N 77 K S R.9 JK 4. JK T T ice N.6JK S W S R S JK S S.6JK R (b) S R JK S.6 JK T T ice N 6 O( c) O(
温泉の化学 1
 H O 1,003 516 149 124 2,237 1974 90 110 1km 2,400 ( 100 Mg 200 (98 ) 43,665 mg 38,695 mg 19,000 mg 2000 2000 Na-Ca-Cl 806 1970 1989 10 1991 4 ph 1 981 10,000 1993... (^^; (SO_4^{2-}) " " 1973-1987 1970
H O 1,003 516 149 124 2,237 1974 90 110 1km 2,400 ( 100 Mg 200 (98 ) 43,665 mg 38,695 mg 19,000 mg 2000 2000 Na-Ca-Cl 806 1970 1989 10 1991 4 ph 1 981 10,000 1993... (^^; (SO_4^{2-}) " " 1973-1987 1970
近畿中国四国農業研究センター研究報告 第7号
 230 C B A D E 50m 558 0 1km (mg L 1 ) T N NO 2 3 N NH 4 N 2.0 0 (a) 2001 1.5 6 20 21 5 1.0 0.5 0.0 2.0 1.5 1.0 0.5 0.0 14:00 17:00 (b) 2001 7 3 4 20:00 23:00 2:00 (h) 5:00 8:00 11:00 10 0 5 10 15
230 C B A D E 50m 558 0 1km (mg L 1 ) T N NO 2 3 N NH 4 N 2.0 0 (a) 2001 1.5 6 20 21 5 1.0 0.5 0.0 2.0 1.5 1.0 0.5 0.0 14:00 17:00 (b) 2001 7 3 4 20:00 23:00 2:00 (h) 5:00 8:00 11:00 10 0 5 10 15
H22環境地球化学4_化学平衡III_ ppt
 1 2 3 2009年度 環境地球化学 大河内 温度上昇による炭酸水の発泡 気泡 温度が高くなると 溶けきれなくなった 二酸化炭素が気泡として出てくる 4 2009年度 環境地球化学 圧力上昇による炭酸水の発泡 栓を開けると 瓶の中の圧力が急激に 小さくなるので 発泡する 大河内 5 CO 2 K H CO 2 H 2 O K H + 1 HCO 3- K 2 H + CO 3 2- (M) [CO
1 2 3 2009年度 環境地球化学 大河内 温度上昇による炭酸水の発泡 気泡 温度が高くなると 溶けきれなくなった 二酸化炭素が気泡として出てくる 4 2009年度 環境地球化学 圧力上昇による炭酸水の発泡 栓を開けると 瓶の中の圧力が急激に 小さくなるので 発泡する 大河内 5 CO 2 K H CO 2 H 2 O K H + 1 HCO 3- K 2 H + CO 3 2- (M) [CO
Contents 1 1. ph ph ph 7 a) ph 9 b) 10 c) ph 11 a) 11 b) 12 c) ph 14 d) 15 ph a) 18 b) / 19 c)
 ph Theory Guide School experiments ph Natural science laws experience ph live ph learn easily Contents 1 1. ph 5 1.1. 5 1.2. ph 6 1.3. ph 7 a) ph 9 b) 10 c) 11 1.4. ph 11 a) 11 b) 12 c) ph 14 d) 15 ph
ph Theory Guide School experiments ph Natural science laws experience ph live ph learn easily Contents 1 1. ph 5 1.1. 5 1.2. ph 6 1.3. ph 7 a) ph 9 b) 10 c) 11 1.4. ph 11 a) 11 b) 12 c) ph 14 d) 15 ph
 48 13 13 13 38 21 13 3 13 3.1 l l l l l l 4.1.1 5mm 0.5mm 5mm 10 500mL 500mL 500mL 20 1 200 / 4 5 6 3000G 20 G1118 R N 2 10-8 GG Rcm Nrpm 1μm 2 3000G 1 /1 4.1.2PCB PCB 5mm 0.5mm 5mm 10 500mL 500mL 500mL
48 13 13 13 38 21 13 3 13 3.1 l l l l l l 4.1.1 5mm 0.5mm 5mm 10 500mL 500mL 500mL 20 1 200 / 4 5 6 3000G 20 G1118 R N 2 10-8 GG Rcm Nrpm 1μm 2 3000G 1 /1 4.1.2PCB PCB 5mm 0.5mm 5mm 10 500mL 500mL 500mL
土壌の観察・実験テキスト −土壌を調べよう!−
 ( ) 2006 7 20 i 21 1962 1969 1987 1992 2005 65 1972 1977 1997 1977 1998 1982 1998 2002 2004 2005 SPP 1999 ii 1 g cm m 6378km ( ) 4.2, 4.3 5.1 7.1 8.1 4.1 7.3, 7.4 7.1 1 2 7.1 8.2 2 5 6 1, 2 2.3 4.2, 4.3
( ) 2006 7 20 i 21 1962 1969 1987 1992 2005 65 1972 1977 1997 1977 1998 1982 1998 2002 2004 2005 SPP 1999 ii 1 g cm m 6378km ( ) 4.2, 4.3 5.1 7.1 8.1 4.1 7.3, 7.4 7.1 1 2 7.1 8.2 2 5 6 1, 2 2.3 4.2, 4.3
2 A B A B A A B Ea 1 51 Ea 1 A B A B B A B B A Ea 2 A B Ea 1 ( )k 1 Ea 1 Ea 2 Arrhenius 53 Ea R T k 1 = χe 1 Ea RT k 2 = χe 2 Ea RT 53 A B A B
 5. A B B A B A B B A A B A B 2 A [A] B [B] 51 v = k[a][b] 51 A B 3 0 273.16 A B A B A B A A [A] 52 v= k[a] 52 A B 55 2 A B A B A A B Ea 1 51 Ea 1 A B A B B A B B A Ea 2 A B Ea 1 ( )k 1 Ea 1 Ea 2 Arrhenius
5. A B B A B A B B A A B A B 2 A [A] B [B] 51 v = k[a][b] 51 A B 3 0 273.16 A B A B A B A A [A] 52 v= k[a] 52 A B 55 2 A B A B A A B Ea 1 51 Ea 1 A B A B B A B B A Ea 2 A B Ea 1 ( )k 1 Ea 1 Ea 2 Arrhenius
Note.tex 2008/09/19( )
 1 20 9 19 2 1 5 1.1........................ 5 1.2............................. 8 2 9 2.1............................. 9 2.2.............................. 10 3 13 3.1.............................. 13 3.2..................................
1 20 9 19 2 1 5 1.1........................ 5 1.2............................. 8 2 9 2.1............................. 9 2.2.............................. 10 3 13 3.1.............................. 13 3.2..................................
1 12 *1 *2 (1991) (1992) (2002) (1991) (1992) (2002) 13 (1991) (1992) (2002) *1 (2003) *2 (1997) 1
 2005 1 1991 1996 5 i 1 12 *1 *2 (1991) (1992) (2002) (1991) (1992) (2002) 13 (1991) (1992) (2002) *1 (2003) *2 (1997) 1 2 13 *3 *4 200 1 14 2 250m :64.3km 457mm :76.4km 200 1 548mm 16 9 12 589 13 8 50m
2005 1 1991 1996 5 i 1 12 *1 *2 (1991) (1992) (2002) (1991) (1992) (2002) 13 (1991) (1992) (2002) *1 (2003) *2 (1997) 1 2 13 *3 *4 200 1 14 2 250m :64.3km 457mm :76.4km 200 1 548mm 16 9 12 589 13 8 50m
/02/18
 3 09/0/8 i III,,,, III,?,,,,,,,,,,,,,,,,,,,,?,?,,,,,,,,,,,,,,!!!,? 3,,,, ii,,,!,,,, OK! :!,,,, :!,,,,,, 3:!,, 4:!,,,, 5:!,,! 7:!,,,,, 8:!,! 9:!,,,,,,,,, ( ),, :, ( ), ( ), 6:!,,, :... : 3 ( )... iii,,
3 09/0/8 i III,,,, III,?,,,,,,,,,,,,,,,,,,,,?,?,,,,,,,,,,,,,,!!!,? 3,,,, ii,,,!,,,, OK! :!,,,, :!,,,,,, 3:!,, 4:!,,,, 5:!,,! 7:!,,,,, 8:!,! 9:!,,,,,,,,, ( ),, :, ( ), ( ), 6:!,,, :... : 3 ( )... iii,,
64 3 g=9.85 m/s 2 g=9.791 m/s 2 36, km ( ) 1 () 2 () m/s : : a) b) kg/m kg/m k
 63 3 Section 3.1 g 3.1 3.1: : 64 3 g=9.85 m/s 2 g=9.791 m/s 2 36, km ( ) 1 () 2 () 3 9.8 m/s 2 3.2 3.2: : a) b) 5 15 4 1 1. 1 3 14. 1 3 kg/m 3 2 3.3 1 3 5.8 1 3 kg/m 3 3 2.65 1 3 kg/m 3 4 6 m 3.1. 65 5
63 3 Section 3.1 g 3.1 3.1: : 64 3 g=9.85 m/s 2 g=9.791 m/s 2 36, km ( ) 1 () 2 () 3 9.8 m/s 2 3.2 3.2: : a) b) 5 15 4 1 1. 1 3 14. 1 3 kg/m 3 2 3.3 1 3 5.8 1 3 kg/m 3 3 2.65 1 3 kg/m 3 4 6 m 3.1. 65 5
1. 4cm 16 cm 4cm 20cm 18 cm L λ(x)=ax [kg/m] A x 4cm A 4cm 12 cm h h Y 0 a G 0.38h a b x r(x) x y = 1 h 0.38h G b h X x r(x) 1 S(x) = πr(x) 2 a,b, h,π
![1. 4cm 16 cm 4cm 20cm 18 cm L λ(x)=ax [kg/m] A x 4cm A 4cm 12 cm h h Y 0 a G 0.38h a b x r(x) x y = 1 h 0.38h G b h X x r(x) 1 S(x) = πr(x) 2 a,b, h,π 1. 4cm 16 cm 4cm 20cm 18 cm L λ(x)=ax [kg/m] A x 4cm A 4cm 12 cm h h Y 0 a G 0.38h a b x r(x) x y = 1 h 0.38h G b h X x r(x) 1 S(x) = πr(x) 2 a,b, h,π](/thumbs/101/149329485.jpg) . 4cm 6 cm 4cm cm 8 cm λ()=a [kg/m] A 4cm A 4cm cm h h Y a G.38h a b () y = h.38h G b h X () S() = π() a,b, h,π V = ρ M = ρv G = M h S() 3 d a,b, h 4 G = 5 h a b a b = 6 ω() s v m θ() m v () θ() ω() dθ()
. 4cm 6 cm 4cm cm 8 cm λ()=a [kg/m] A 4cm A 4cm cm h h Y a G.38h a b () y = h.38h G b h X () S() = π() a,b, h,π V = ρ M = ρv G = M h S() 3 d a,b, h 4 G = 5 h a b a b = 6 ω() s v m θ() m v () θ() ω() dθ()
第4次酸性雨全国調査報告書(平成16年度) -(Ⅱ)付表編-
 1.12.24 P. 5.4.3.1 NO NOP. 5.4.3.2P. 5.4.3.3 P. 5.4.3.4 P. 2.18 2.19 P. 2.20 P. 2.21 P. P. 5.5.2.11 SOA P. 5.5.2.12 NO B A B 1.1 16 mm FRS Vol. 31 42006 1.2 ph 16 FRS ms/m FRS 1.3 16 Vol. 31 42006 μmol/l
1.12.24 P. 5.4.3.1 NO NOP. 5.4.3.2P. 5.4.3.3 P. 5.4.3.4 P. 2.18 2.19 P. 2.20 P. 2.21 P. P. 5.5.2.11 SOA P. 5.5.2.12 NO B A B 1.1 16 mm FRS Vol. 31 42006 1.2 ph 16 FRS ms/m FRS 1.3 16 Vol. 31 42006 μmol/l
PowerPoint プレゼンテーション
 ICP-AES, AA ppb - 1% ICP-MS ppt - 100ppm SEM-EDX 0.1% - 100% B - U XRF 0.01% - 100% Na - U EMIA ppm - 100% C, S EMGA EMIA : EMGA ppm - 100% O, N, H Carbon Sulfur Nitrigen Oxygen ISO ISO/TC 17/SC ISO 9556
ICP-AES, AA ppb - 1% ICP-MS ppt - 100ppm SEM-EDX 0.1% - 100% B - U XRF 0.01% - 100% Na - U EMIA ppm - 100% C, S EMGA EMIA : EMGA ppm - 100% O, N, H Carbon Sulfur Nitrigen Oxygen ISO ISO/TC 17/SC ISO 9556
 ver 0.3 Chapter 0 0.1 () 0( ) 0.2 3 4 CHAPTER 0. http://www.jaist.ac.jp/~t-yama/k116 0.3 50% ( Wikipedia ) ( ) 0.4! 2006 0.4. 5 MIT OCW ( ) MIT Open Courseware MIT (Massachusetts Institute of Technology)
ver 0.3 Chapter 0 0.1 () 0( ) 0.2 3 4 CHAPTER 0. http://www.jaist.ac.jp/~t-yama/k116 0.3 50% ( Wikipedia ) ( ) 0.4! 2006 0.4. 5 MIT OCW ( ) MIT Open Courseware MIT (Massachusetts Institute of Technology)
4. 5.
 1. 2. 20 3. 4. 5. ) N ) 6. ) 7. 8. JA 1. 30 CO2 18 2. 20 3. 4. 5. 5.1 cm 5.2 5.2.1 1 17kg 119kg11 5.2.2 60 750 2 2 5.2.3 30m 5.2.4 3 200g 6. 1 500 200 3 2 16 600 21 /10067kg/198kg10037 2025 3 4 3 3 6 102
1. 2. 20 3. 4. 5. ) N ) 6. ) 7. 8. JA 1. 30 CO2 18 2. 20 3. 4. 5. 5.1 cm 5.2 5.2.1 1 17kg 119kg11 5.2.2 60 750 2 2 5.2.3 30m 5.2.4 3 200g 6. 1 500 200 3 2 16 600 21 /10067kg/198kg10037 2025 3 4 3 3 6 102
V(x) m e V 0 cos x π x π V(x) = x < π, x > π V 0 (i) x = 0 (V(x) V 0 (1 x 2 /2)) n n d 2 f dξ 2ξ d f 2 dξ + 2n f = 0 H n (ξ) (ii) H
 199 1 1 199 1 1. Vx) m e V cos x π x π Vx) = x < π, x > π V i) x = Vx) V 1 x /)) n n d f dξ ξ d f dξ + n f = H n ξ) ii) H n ξ) = 1) n expξ ) dn dξ n exp ξ )) H n ξ)h m ξ) exp ξ )dξ = π n n!δ n,m x = Vx)
199 1 1 199 1 1. Vx) m e V cos x π x π Vx) = x < π, x > π V i) x = Vx) V 1 x /)) n n d f dξ ξ d f dξ + n f = H n ξ) ii) H n ξ) = 1) n expξ ) dn dξ n exp ξ )) H n ξ)h m ξ) exp ξ )dξ = π n n!δ n,m x = Vx)
n 2 + π2 6 x [10 n x] x = lim n 10 n n 10 k x 1.1. a 1, a 2,, a n, (a n ) n=1 {a n } n=1 1.2 ( ). {a n } n=1 Q ε > 0 N N m, n N a m
![n 2 + π2 6 x [10 n x] x = lim n 10 n n 10 k x 1.1. a 1, a 2,, a n, (a n ) n=1 {a n } n=1 1.2 ( ). {a n } n=1 Q ε > 0 N N m, n N a m n 2 + π2 6 x [10 n x] x = lim n 10 n n 10 k x 1.1. a 1, a 2,, a n, (a n ) n=1 {a n } n=1 1.2 ( ). {a n } n=1 Q ε > 0 N N m, n N a m](/thumbs/94/118912662.jpg) 1 1 1 + 1 4 + + 1 n 2 + π2 6 x [10 n x] x = lim n 10 n n 10 k x 1.1. a 1, a 2,, a n, (a n ) n=1 {a n } n=1 1.2 ( ). {a n } n=1 Q ε > 0 N N m, n N a m a n < ε 1 1. ε = 10 1 N m, n N a m a n < ε = 10 1 N
1 1 1 + 1 4 + + 1 n 2 + π2 6 x [10 n x] x = lim n 10 n n 10 k x 1.1. a 1, a 2,, a n, (a n ) n=1 {a n } n=1 1.2 ( ). {a n } n=1 Q ε > 0 N N m, n N a m a n < ε 1 1. ε = 10 1 N m, n N a m a n < ε = 10 1 N
(1.2) T D = 0 T = D = 30 kn 1.2 (1.4) 2F W = 0 F = W/2 = 300 kn/2 = 150 kn 1.3 (1.9) R = W 1 + W 2 = = 1100 N. (1.9) W 2 b W 1 a = 0
 1 1 1.1 1.) T D = T = D = kn 1. 1.4) F W = F = W/ = kn/ = 15 kn 1. 1.9) R = W 1 + W = 6 + 5 = 11 N. 1.9) W b W 1 a = a = W /W 1 )b = 5/6) = 5 cm 1.4 AB AC P 1, P x, y x, y y x 1.4.) P sin 6 + P 1 sin 45
1 1 1.1 1.) T D = T = D = kn 1. 1.4) F W = F = W/ = kn/ = 15 kn 1. 1.9) R = W 1 + W = 6 + 5 = 11 N. 1.9) W b W 1 a = a = W /W 1 )b = 5/6) = 5 cm 1.4 AB AC P 1, P x, y x, y y x 1.4.) P sin 6 + P 1 sin 45
物理化学I-第12回(13).ppt
 I- 12-1 11 11.1 2Mg(s) + O 2 (g) 2MgO(s) [Mg 2+ O 2 ] Zn(s) + Cu 2+ (aq) Zn 2+ (aq) + Cu(s) - 2Mg(s) 2Mg 2+ (s) + 4e +) O 2 (g) + 4e 2O 2 (s) 2Mg(s) + O 2 (g) 2MgO(s) Zn(s) Zn 2+ (aq) + 2e +) Cu 2+ (aq)
I- 12-1 11 11.1 2Mg(s) + O 2 (g) 2MgO(s) [Mg 2+ O 2 ] Zn(s) + Cu 2+ (aq) Zn 2+ (aq) + Cu(s) - 2Mg(s) 2Mg 2+ (s) + 4e +) O 2 (g) + 4e 2O 2 (s) 2Mg(s) + O 2 (g) 2MgO(s) Zn(s) Zn 2+ (aq) + 2e +) Cu 2+ (aq)
1
 I II II 1 dw = pd = 0 1 U = Q (4.10) 1K (heat capacity) (mole heat capacity) ( dq / d ) = ( du d C = / ) (4.11) du = C d U = C d (4.1) 1 1 du = dq + dw dw = pd dq = du + pd (4.13) p dq = d( U + p ) p (4.14)
I II II 1 dw = pd = 0 1 U = Q (4.10) 1K (heat capacity) (mole heat capacity) ( dq / d ) = ( du d C = / ) (4.11) du = C d U = C d (4.1) 1 1 du = dq + dw dw = pd dq = du + pd (4.13) p dq = d( U + p ) p (4.14)
untitled
 NPO 2006( ) 11 14 ( ) (2006/12/3) 1 50% % - - (CO+H2) ( ) 6 44 1) --- 2) ( CO H2 ) 2 3 3 90 3 3 2 3 2004 ( ) 1 1 4 1 20% 5 ( ) ( ) 2 6 MAWERA ) MAWERA ( ) ( ) 7 6MW -- 175kW 8 ( ) 900 10 2 2 2 9 -- - 10
NPO 2006( ) 11 14 ( ) (2006/12/3) 1 50% % - - (CO+H2) ( ) 6 44 1) --- 2) ( CO H2 ) 2 3 3 90 3 3 2 3 2004 ( ) 1 1 4 1 20% 5 ( ) ( ) 2 6 MAWERA ) MAWERA ( ) ( ) 7 6MW -- 175kW 8 ( ) 900 10 2 2 2 9 -- - 10
RAA-05(201604)MRA対応製品ver6
 M R A 対 応 製 品 ISO/IEC 17025 ISO/IEC 17025は 試験所及び校正機関が特定の試験又は 校正を実施する能力があるものとして認定を 受けようとする場合の一般要求事項を規定した国際規格 国際相互承認 MRA Mutual Recognition Arrangement 相互承認協定 とは 試験 検査を実施する試験所 検査機関を認定する国際組織として ILAC 国際試験所認定協力機構
M R A 対 応 製 品 ISO/IEC 17025 ISO/IEC 17025は 試験所及び校正機関が特定の試験又は 校正を実施する能力があるものとして認定を 受けようとする場合の一般要求事項を規定した国際規格 国際相互承認 MRA Mutual Recognition Arrangement 相互承認協定 とは 試験 検査を実施する試験所 検査機関を認定する国際組織として ILAC 国際試験所認定協力機構
<4D F736F F F696E74202D208D918E8E91CE8DF481698E5F89968AEE816A F38DFC97702E707074>
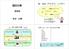 国試対策 酸塩基 酸 塩基 ( アルカリ ) って何? H 2 SO 4 水酸化カルシウムアンモニア CH HNO 3 4 HCl 硫酸ナトリウム 新垣 知輝 どれが酸? どれが塩基? 酸 酸と塩基の定義 塩基 H + を出すもの OH を出すもの by アレニウス H + を渡すもの H + を受け取るもの by ブレンステッド ローリー電子対を受け取る電子対を渡すものもの by ルイス 水素イオン
国試対策 酸塩基 酸 塩基 ( アルカリ ) って何? H 2 SO 4 水酸化カルシウムアンモニア CH HNO 3 4 HCl 硫酸ナトリウム 新垣 知輝 どれが酸? どれが塩基? 酸 酸と塩基の定義 塩基 H + を出すもの OH を出すもの by アレニウス H + を渡すもの H + を受け取るもの by ブレンステッド ローリー電子対を受け取る電子対を渡すものもの by ルイス 水素イオン
QMI_10.dvi
 ... black body radiation black body black body radiation Gustav Kirchhoff 859 895 W. Wien O.R. Lummer cavity radiation ν ν +dν f T (ν) f T (ν)dν = 8πν2 c 3 kt dν (Rayleigh Jeans) (.) f T (ν) spectral energy
... black body radiation black body black body radiation Gustav Kirchhoff 859 895 W. Wien O.R. Lummer cavity radiation ν ν +dν f T (ν) f T (ν)dν = 8πν2 c 3 kt dν (Rayleigh Jeans) (.) f T (ν) spectral energy
 8 i, III,,,, III,, :!,,,, :!,,,,, 4:!,,,,,,!,,,, OK! 5:!,,,,,,,,,, OK 6:!, 0, 3:!,,,,! 7:!,,,,,, ii,,,,,, ( ),, :, ( ), ( ), :... : 3 ( )...,, () : ( )..., :,,, ( ), (,,, ),, (ϵ δ ), ( ), (ˆ ˆ;),,,,,,!,,,,.,,
8 i, III,,,, III,, :!,,,, :!,,,,, 4:!,,,,,,!,,,, OK! 5:!,,,,,,,,,, OK 6:!, 0, 3:!,,,,! 7:!,,,,,, ii,,,,,, ( ),, :, ( ), ( ), :... : 3 ( )...,, () : ( )..., :,,, ( ), (,,, ),, (ϵ δ ), ( ), (ˆ ˆ;),,,,,,!,,,,.,,
Part () () Γ Part ,
 Contents a 6 6 6 6 6 6 6 7 7. 8.. 8.. 8.3. 8 Part. 9. 9.. 9.. 3. 3.. 3.. 3 4. 5 4.. 5 4.. 9 4.3. 3 Part. 6 5. () 6 5.. () 7 5.. 9 5.3. Γ 3 6. 3 6.. 3 6.. 3 6.3. 33 Part 3. 34 7. 34 7.. 34 7.. 34 8. 35
Contents a 6 6 6 6 6 6 6 7 7. 8.. 8.. 8.3. 8 Part. 9. 9.. 9.. 3. 3.. 3.. 3 4. 5 4.. 5 4.. 9 4.3. 3 Part. 6 5. () 6 5.. () 7 5.. 9 5.3. Γ 3 6. 3 6.. 3 6.. 3 6.3. 33 Part 3. 34 7. 34 7.. 34 7.. 34 8. 35
 http://www.ike-dyn.ritsumei.ac.jp/ hyoo/wave.html 1 1, 5 3 1.1 1..................................... 3 1.2 5.1................................... 4 1.3.......................... 5 1.4 5.2, 5.3....................
http://www.ike-dyn.ritsumei.ac.jp/ hyoo/wave.html 1 1, 5 3 1.1 1..................................... 3 1.2 5.1................................... 4 1.3.......................... 5 1.4 5.2, 5.3....................
untitled
 ( ) c a sin b c b c a cos a c b c a tan b a b cos sin a c b c a ccos b csin (4) Ma k Mg a (Gal) g(98gal) (Gal) a max (K-E) kh Zck.85.6. 4 Ma g a k a g k D τ f c + σ tanφ σ 3 3 /A τ f3 S S τ A σ /A σ /A
( ) c a sin b c b c a cos a c b c a tan b a b cos sin a c b c a ccos b csin (4) Ma k Mg a (Gal) g(98gal) (Gal) a max (K-E) kh Zck.85.6. 4 Ma g a k a g k D τ f c + σ tanφ σ 3 3 /A τ f3 S S τ A σ /A σ /A
36 th IChO : - 3 ( ) , G O O D L U C K final 1
 36 th ICh - - 5 - - : - 3 ( ) - 169 - -, - - - - - - - G D L U C K final 1 1 1.01 2 e 4.00 3 Li 6.94 4 Be 9.01 5 B 10.81 6 C 12.01 7 N 14.01 8 16.00 9 F 19.00 10 Ne 20.18 11 Na 22.99 12 Mg 24.31 Periodic
36 th ICh - - 5 - - : - 3 ( ) - 169 - -, - - - - - - - G D L U C K final 1 1 1.01 2 e 4.00 3 Li 6.94 4 Be 9.01 5 B 10.81 6 C 12.01 7 N 14.01 8 16.00 9 F 19.00 10 Ne 20.18 11 Na 22.99 12 Mg 24.31 Periodic
4. ϵ(ν, T ) = c 4 u(ν, T ) ϵ(ν, T ) T ν π4 Planck dx = 0 e x 1 15 U(T ) x 3 U(T ) = σt 4 Stefan-Boltzmann σ 2π5 k 4 15c 2 h 3 = W m 2 K 4 5.
 A 1. Boltzmann Planck u(ν, T )dν = 8πh ν 3 c 3 kt 1 dν h 6.63 10 34 J s Planck k 1.38 10 23 J K 1 Boltzmann u(ν, T ) T ν e hν c = 3 10 8 m s 1 2. Planck λ = c/ν Rayleigh-Jeans u(ν, T )dν = 8πν2 kt dν c
A 1. Boltzmann Planck u(ν, T )dν = 8πh ν 3 c 3 kt 1 dν h 6.63 10 34 J s Planck k 1.38 10 23 J K 1 Boltzmann u(ν, T ) T ν e hν c = 3 10 8 m s 1 2. Planck λ = c/ν Rayleigh-Jeans u(ν, T )dν = 8πν2 kt dν c
 2010 1 1 7 2 16 2 2 06in 3 3 錆 錆 06in 4 4 0.1 5 5 HCl Cl NaOH H Cl O 2 H H 2 O NaCl Na OH H 2 O NaCl 2010 6 6 ph3.0 30 mg/l 30 1 O-157 H7 2010 7 7 100 Cl 80 60 40 20 HClO ClO CL ph 10 100,000ppm 12 01
2010 1 1 7 2 16 2 2 06in 3 3 錆 錆 06in 4 4 0.1 5 5 HCl Cl NaOH H Cl O 2 H H 2 O NaCl Na OH H 2 O NaCl 2010 6 6 ph3.0 30 mg/l 30 1 O-157 H7 2010 7 7 100 Cl 80 60 40 20 HClO ClO CL ph 10 100,000ppm 12 01
PowerPoint プレゼンテーション
 003.10.3 003.10.8 Y 1 0031016 B4(4 3 B4,1 M 0 C,Q 0. M,Q 1.- MQ 003/10/16 10/8 Girder BeamColumn Foundation SlabWall Girder BeamColumn Foundation SlabWall 1.-1 5mm 0 kn/m 3 0.05m=0.5 kn/m 60mm 18 kn/m
003.10.3 003.10.8 Y 1 0031016 B4(4 3 B4,1 M 0 C,Q 0. M,Q 1.- MQ 003/10/16 10/8 Girder BeamColumn Foundation SlabWall Girder BeamColumn Foundation SlabWall 1.-1 5mm 0 kn/m 3 0.05m=0.5 kn/m 60mm 18 kn/m
20 57
 56 20 57 58 59 12 60 ph 61 62 CCA 63 64 ( 1) 700 7.5 86% 54 17 71 2) 700 6.8 86% 50 15 65 3) 600 700 5.7 84% 36 14 50 550 650 4.2 80% 21 6 27 40 500 3.6 78% 16 7 23 3.6 78% 16 7 23 400 700 4.0 78% 18 7
56 20 57 58 59 12 60 ph 61 62 CCA 63 64 ( 1) 700 7.5 86% 54 17 71 2) 700 6.8 86% 50 15 65 3) 600 700 5.7 84% 36 14 50 550 650 4.2 80% 21 6 27 40 500 3.6 78% 16 7 23 3.6 78% 16 7 23 400 700 4.0 78% 18 7
 ッ ー ー ー() () 2.5 () () 21 1) -1 2) -2 10 m 5 7 23 6 1)2) 3) -1-2 H23.4.25 -15-20 4 75 5 1) -1 (5) (3) (3) (2) (1) (1) (1) (1) (1) (1) (1) (1) (1) (1) (1) (1) 1) ( 23 6 8 ) 2) 22 3)
ッ ー ー ー() () 2.5 () () 21 1) -1 2) -2 10 m 5 7 23 6 1)2) 3) -1-2 H23.4.25 -15-20 4 75 5 1) -1 (5) (3) (3) (2) (1) (1) (1) (1) (1) (1) (1) (1) (1) (1) (1) (1) 1) ( 23 6 8 ) 2) 22 3)
y = x x R = 0. 9, R = σ $ = y x w = x y x x w = x y α ε = + β + x x x y α ε = + β + γ x + x x x x' = / x y' = y/ x y' =
 y x = α + β + ε =,, ε V( ε) = E( ε ) = σ α $ $ β w ( 0) σ = w σ σ y α x ε = + β + w w w w ε / w ( w y x α β ) = α$ $ W = yw βwxw $β = W ( W) ( W)( W) w x x w x x y y = = x W y W x y x y xw = y W = w w
y x = α + β + ε =,, ε V( ε) = E( ε ) = σ α $ $ β w ( 0) σ = w σ σ y α x ε = + β + w w w w ε / w ( w y x α β ) = α$ $ W = yw βwxw $β = W ( W) ( W)( W) w x x w x x y y = = x W y W x y x y xw = y W = w w
CRA3689A
 AVIC-DRZ90 AVIC-DRZ80 2 3 4 5 66 7 88 9 10 10 10 11 12 13 14 15 1 1 0 OPEN ANGLE REMOTE WIDE SET UP AVIC-DRZ90 SOURCE OFF AV CONTROL MIC 2 16 17 1 2 0 0 1 AVIC-DRZ90 2 3 4 OPEN ANGLE REMOTE SOURCE OFF
AVIC-DRZ90 AVIC-DRZ80 2 3 4 5 66 7 88 9 10 10 10 11 12 13 14 15 1 1 0 OPEN ANGLE REMOTE WIDE SET UP AVIC-DRZ90 SOURCE OFF AV CONTROL MIC 2 16 17 1 2 0 0 1 AVIC-DRZ90 2 3 4 OPEN ANGLE REMOTE SOURCE OFF
<4D F736F F D B B83578B6594BB2D834A836F815B82D082C88C602E646F63>
 スピントロニクスの基礎 サンプルページ この本の定価 判型などは, 以下の URL からご覧いただけます. http://www.morikita.co.jp/books/mid/077461 このサンプルページの内容は, 初版 1 刷発行時のものです. i 1 2 ii 3 5 4 AMR (anisotropic magnetoresistance effect) GMR (giant magnetoresistance
スピントロニクスの基礎 サンプルページ この本の定価 判型などは, 以下の URL からご覧いただけます. http://www.morikita.co.jp/books/mid/077461 このサンプルページの内容は, 初版 1 刷発行時のものです. i 1 2 ii 3 5 4 AMR (anisotropic magnetoresistance effect) GMR (giant magnetoresistance
<4D F736F F D B BA908593B98AC782AB82E593E082CC88C091538AC7979D82C98AD682B782E992868AD495F18D908F912E646F63>
 12 62 1800 14 3 11 14 4 14 4 30 ... 1... 1... 1... 2... 5... 6... 7... 10... 13... 18... 19 21 22 24 27 28 31 32 33... 34... 35... 37 2 1,650mm () H=2,500W=2,000 460m 750m 500m 42.1m 1.15m () 100% 80%
12 62 1800 14 3 11 14 4 14 4 30 ... 1... 1... 1... 2... 5... 6... 7... 10... 13... 18... 19 21 22 24 27 28 31 32 33... 34... 35... 37 2 1,650mm () H=2,500W=2,000 460m 750m 500m 42.1m 1.15m () 100% 80%
ナノ粒子のサイズ・形態制御と 構造敏感型触媒プロセスへの応用
 2 3 10 9 m = 1 nm m 4 5 100 m 1m 10cm 100 m 1cm 1mm 10 m 1 m 100nm 10nm 1nm 1 10 m 1 m 1nm 100nm 10nm 10 8 6 1 nm 10 8 7 9 10 11 12 13 14 15 17 18 19 20 21 nm Ni-Zn 2- Ni-Zn Zn Ni Ni-Zn B 5 10 nm 23
2 3 10 9 m = 1 nm m 4 5 100 m 1m 10cm 100 m 1cm 1mm 10 m 1 m 100nm 10nm 1nm 1 10 m 1 m 1nm 100nm 10nm 10 8 6 1 nm 10 8 7 9 10 11 12 13 14 15 17 18 19 20 21 nm Ni-Zn 2- Ni-Zn Zn Ni Ni-Zn B 5 10 nm 23
untitled
 8- My + Cy + Ky = f () t 8. C f () t ( t) = Ψq( t) () t = Ψq () t () t = Ψq () t = ( q q ) ; = [ ] y y y q Ψ φ φ φ = ( ϕ, ϕ, ϕ,3 ) 8. ψ Ψ MΨq + Ψ CΨq + Ψ KΨq = Ψ f ( t) Ψ MΨ = I; Ψ CΨ = C; Ψ KΨ = Λ; q
8- My + Cy + Ky = f () t 8. C f () t ( t) = Ψq( t) () t = Ψq () t () t = Ψq () t = ( q q ) ; = [ ] y y y q Ψ φ φ φ = ( ϕ, ϕ, ϕ,3 ) 8. ψ Ψ MΨq + Ψ CΨq + Ψ KΨq = Ψ f ( t) Ψ MΨ = I; Ψ CΨ = C; Ψ KΨ = Λ; q
% %
 4 8 4 5 414.5 172.1 41.5% 39.8 10% 17 186.4 70.8 4 9 10 11 3 1-1 - 2-2 - - 3 - 10% - 4 - 4060% 5560% 60% 40% 3040 1520 1015 411 7 4 11 610 7080 5060 100 3040 13-5 - 1015 4045 3.04.0 5.06.0 1.52.0 1.01.5
4 8 4 5 414.5 172.1 41.5% 39.8 10% 17 186.4 70.8 4 9 10 11 3 1-1 - 2-2 - - 3 - 10% - 4 - 4060% 5560% 60% 40% 3040 1520 1015 411 7 4 11 610 7080 5060 100 3040 13-5 - 1015 4045 3.04.0 5.06.0 1.52.0 1.01.5
研修コーナー
 l l l l l l l l l l l α α β l µ l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l
l l l l l l l l l l l α α β l µ l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l l
1.500 m X Y m m m m m m m m m m m m N/ N/ ( ) qa N/ N/ 2 2
 1.500 m X Y 0.200 m 0.200 m 0.200 m 0.200 m 0.200 m 0.000 m 1.200 m m 0.150 m 0.150 m m m 2 24.5 N/ 3 18.0 N/ 3 30.0 0.60 ( ) qa 50.79 N/ 2 0.0 N/ 2 20.000 20.000 15.000 15.000 X(m) Y(m) (kn/m 2 ) 10.000
1.500 m X Y 0.200 m 0.200 m 0.200 m 0.200 m 0.200 m 0.000 m 1.200 m m 0.150 m 0.150 m m m 2 24.5 N/ 3 18.0 N/ 3 30.0 0.60 ( ) qa 50.79 N/ 2 0.0 N/ 2 20.000 20.000 15.000 15.000 X(m) Y(m) (kn/m 2 ) 10.000
CuSO POINT S 2 Ni Sn Hg Cu Ag Zn 2 Cu Cu Cu OH 2 Cu NH CuSO 4 5H 2O Ag Ag 2O Ag 2CrO4 Zn ZnS ZnO 2+ Fe Fe OH 2 Fe 3+ Fe OH 3 2 Cu Cu OH 2 Ag Ag
 CuSO POINT S 2 Ni Sn Hg Cu Ag Zn 2 CuCu Cu OH 2 Cu NH 3 4 2 CuSO 4 5H 2O AgAg 2O Ag 2CrO4 ZnZnS ZnO 2+ Fe Fe OH 2 Fe 3+ Fe OH 3 2 Cu Cu OH 2 Ag Ag 2O Cl Cl AgCl PbCl 2 Ag Cl AgCl Pb 2 2Cl PbCl2 Cl Hg22
CuSO POINT S 2 Ni Sn Hg Cu Ag Zn 2 CuCu Cu OH 2 Cu NH 3 4 2 CuSO 4 5H 2O AgAg 2O Ag 2CrO4 ZnZnS ZnO 2+ Fe Fe OH 2 Fe 3+ Fe OH 3 2 Cu Cu OH 2 Ag Ag 2O Cl Cl AgCl PbCl 2 Ag Cl AgCl Pb 2 2Cl PbCl2 Cl Hg22
) km 200 m ) ) ) ) ) ) ) kg kg ) 017 x y x 2 y 5x 5 y )
 001 ) g 20 g 5 300 g 7 002 720 g 2 ) g 003 0.8 m 2 ) cm 2 004 12 15 1 3 1 ) 005 5 0.8 0.4 ) 6 006 5 2 3 66 ) 007 1 700 12 ) 008 0.315 ) 009 500 g ) kg 0.2 t 189 kg 17.1 kg 010 5 1 2 cm 3 cm )km 2-1 - 011
001 ) g 20 g 5 300 g 7 002 720 g 2 ) g 003 0.8 m 2 ) cm 2 004 12 15 1 3 1 ) 005 5 0.8 0.4 ) 6 006 5 2 3 66 ) 007 1 700 12 ) 008 0.315 ) 009 500 g ) kg 0.2 t 189 kg 17.1 kg 010 5 1 2 cm 3 cm )km 2-1 - 011
NETES No.CG V
 1 2006 6 NETES No.CG-050001-V 2007 5 2 1 2 1 19 5 1 2 19 8 2 i 1 1 1.1 1 1.2 2 1.3 2 2 3 2.1 3 2.2 8 3 9 3.1 9 3.2 10 3.3 13 3.3.1 13 3.3.2 14 3.3.3 14 3.3.4 16 3.3.5 17 3.3.6 18 3.3.7 21 3.3.8 22 3.4
1 2006 6 NETES No.CG-050001-V 2007 5 2 1 2 1 19 5 1 2 19 8 2 i 1 1 1.1 1 1.2 2 1.3 2 2 3 2.1 3 2.2 8 3 9 3.1 9 3.2 10 3.3 13 3.3.1 13 3.3.2 14 3.3.3 14 3.3.4 16 3.3.5 17 3.3.6 18 3.3.7 21 3.3.8 22 3.4
第十四改正日本薬局方第二追補
 461 3 3 3 5 5 6 6 10 18 19 79 81 101 113 127 133 139 145 147 152 153 156 158 165 3 19 20 21 22 22 23 24 25 26 26 27 28 29 29 30 31 dl 32 dl 33 34 34 36 36 37 38 38 38 39 40 42 42 42 43 44 45 46 46 47 48
461 3 3 3 5 5 6 6 10 18 19 79 81 101 113 127 133 139 145 147 152 153 156 158 165 3 19 20 21 22 22 23 24 25 26 26 27 28 29 29 30 31 dl 32 dl 33 34 34 36 36 37 38 38 38 39 40 42 42 42 43 44 45 46 46 47 48
N cos s s cos ψ e e e e 3 3 e e 3 e 3 e
 3 3 5 5 5 3 3 7 5 33 5 33 9 5 8 > e > f U f U u u > u ue u e u ue u ue u e u e u u e u u e u N cos s s cos ψ e e e e 3 3 e e 3 e 3 e 3 > A A > A E A f A A f A [ ] f A A e > > A e[ ] > f A E A < < f ; >
3 3 5 5 5 3 3 7 5 33 5 33 9 5 8 > e > f U f U u u > u ue u e u ue u ue u e u e u u e u u e u N cos s s cos ψ e e e e 3 3 e e 3 e 3 e 3 > A A > A E A f A A f A [ ] f A A e > > A e[ ] > f A E A < < f ; >
.5 z = a + b + c n.6 = a sin t y = b cos t dy d a e e b e + e c e e e + e 3 s36 3 a + y = a, b > b 3 s363.7 y = + 3 y = + 3 s364.8 cos a 3 s365.9 y =,
 [ ] IC. r, θ r, θ π, y y = 3 3 = r cos θ r sin θ D D = {, y ; y }, y D r, θ ep y yddy D D 9 s96. d y dt + 3dy + y = cos t dt t = y = e π + e π +. t = π y =.9 s6.3 d y d + dy d + y = y =, dy d = 3 a, b
[ ] IC. r, θ r, θ π, y y = 3 3 = r cos θ r sin θ D D = {, y ; y }, y D r, θ ep y yddy D D 9 s96. d y dt + 3dy + y = cos t dt t = y = e π + e π +. t = π y =.9 s6.3 d y d + dy d + y = y =, dy d = 3 a, b
I-2 (100 ) (1) y(x) y dy dx y d2 y dx 2 (a) y + 2y 3y = 9e 2x (b) x 2 y 6y = 5x 4 (2) Bernoulli B n (n = 0, 1, 2,...) x e x 1 = n=0 B 0 B 1 B 2 (3) co
 16 I ( ) (1) I-1 I-2 I-3 (2) I-1 ( ) (100 ) 2l x x = 0 y t y(x, t) y(±l, t) = 0 m T g y(x, t) l y(x, t) c = 2 y(x, t) c 2 2 y(x, t) = g (A) t 2 x 2 T/m (1) y 0 (x) y 0 (x) = g c 2 (l2 x 2 ) (B) (2) (1)
16 I ( ) (1) I-1 I-2 I-3 (2) I-1 ( ) (100 ) 2l x x = 0 y t y(x, t) y(±l, t) = 0 m T g y(x, t) l y(x, t) c = 2 y(x, t) c 2 2 y(x, t) = g (A) t 2 x 2 T/m (1) y 0 (x) y 0 (x) = g c 2 (l2 x 2 ) (B) (2) (1)
1 9 v.0.1 c (2016/10/07) Minoru Suzuki T µ 1 (7.108) f(e ) = 1 e β(e µ) 1 E 1 f(e ) (Bose-Einstein distribution function) *1 (8.1) (9.1)
 1 9 v..1 c (216/1/7) Minoru Suzuki 1 1 9.1 9.1.1 T µ 1 (7.18) f(e ) = 1 e β(e µ) 1 E 1 f(e ) (Bose-Einstein distribution function) *1 (8.1) (9.1) E E µ = E f(e ) E µ (9.1) µ (9.2) µ 1 e β(e µ) 1 f(e )
1 9 v..1 c (216/1/7) Minoru Suzuki 1 1 9.1 9.1.1 T µ 1 (7.18) f(e ) = 1 e β(e µ) 1 E 1 f(e ) (Bose-Einstein distribution function) *1 (8.1) (9.1) E E µ = E f(e ) E µ (9.1) µ (9.2) µ 1 e β(e µ) 1 f(e )
(a) (b) X Ag + + X AgX F < Cl < Br < I Li + + X LiX F > Cl > Br > I (a) (b) (c)
 ( 13 : 30 16 : 00 ) (a) (b) X Ag + + X AgX F < Cl < Br < I Li + + X LiX F > Cl > Br > I (a) (b) (c) (a) CH 3 -Br (b) (c),2,4- (d) CH 3 O-CH=CH-CH 2 (a) NH 2 CH 3 H 3 C NH 2 H CH 3 CH 3 NH 2 H 3 C CH 3
( 13 : 30 16 : 00 ) (a) (b) X Ag + + X AgX F < Cl < Br < I Li + + X LiX F > Cl > Br > I (a) (b) (c) (a) CH 3 -Br (b) (c),2,4- (d) CH 3 O-CH=CH-CH 2 (a) NH 2 CH 3 H 3 C NH 2 H CH 3 CH 3 NH 2 H 3 C CH 3










