日本化学療法学会雑誌第56巻第1号
|
|
|
- あつとし こいたばし
- 7 years ago
- Views:
Transcription
1 β β β β β Streptococcus pneumoniaehaemophilus influenzaemoraxella catarrhalismycoplasma pneumoniaechlamydia pneumoniae β Key wordsβ
2 mys nilc laci ngis Table. Assessmentschedule Parameters Patientcharacteristics potm /s s QOL Bodytemperature Sputum volume Sputum color Cough Bacteriologicalexamination Mycoplasma/ Chlamydiaantibodytiter Viralantigentests Clinicallaboratorytests Adverseevents Atinitiationof studytreatment Acuteexacerbationphase Day Day7 Typicaly,onadailybasis Day :Assessmentbytheinvestigator :Assessmentbythepatient :Donewheneverpossible :Assessedandrecordedinthe TreatmentDiary bythepatientoncedaily Re-treatment Stable phase Relapse phase β I β β β
3 Mycoplasma pneumoniae Chlamydia pneumoniae Mycoplasma pneumoniaechlamydia pneumoniae M. pneumoniae C. pneumoniae II β β β β β
4 Patients enrolled: N8 (LVFX: N8, -lactam: N9) Patients evaluated for safety: N8 (LVFX: N96, -lactam: N88) Patients evaluated for efficacy: N9 (LVFX: N9, -lactam: N58) Patients evaluated for QOL: N7 (LVFX: N67, -lactam: N5) Patients excluded from all analyses Patients enrolled outside the study contract period Patients with missing case record cards Patients who used an incorrect antibacterial agent Patients lost to follow-up Patients excluded from efficacy analysis Patients deviating from the eligibility criteria Patients without a Treatment Diary Patients excluded from QOL analysis Patients with missing data for QOL assessment LVFX -lactam Total LVFX -lactam Total LVFX -lactam Total 8 Fig.. Dispositionofthepatients. β β β β β β β β β β β β β β
5 Table. Baselinecharacteristicsofthepatients Gender Variables Male Female Total(%) (85.5) 6(.5) LVFX(%) 6(85.) 8(.7) β -lactam (%) 5(86.) 8(.8) < 65 (7.) 7(9.) 6(.) Age >_ 65< 75 95(8.) 75(9.) (.5) >_ 75 (.6) 79(.) (55.) Height Weight Mean±SD Mean±SD 6.6±7. 5.± ±7. 5.±8.5 6.± ±. Outpatient (89.9) 69(88.9) 5(9.) Inpatient/outpatient Inpatient 5(.) (.) (6.9) Unknown No (.) 8(6.) (7.7) Historyofsmoking Yes 9(85.8) 5(8.8) 8 (9.) Unknown 8 6 Smokingindex Severityoftheinfection (accordingtotheinvestigator) Severityoftheinfection (accordingtotheseverity CriteriaoftheJapanese SocietyofChemotherapy) SeverityofCOPD DurationofCOPD(years) Concomitantdrugs Antibacterialmedication within7dayspriortostudy treatment Lowdoselong-term macrolide therapy Concurentdisease Mean±SD Mild Moderate Severe Unknown Mild Moderate Severe Unknown Mild Moderate Severe VerySevere Unknown Mean±SD No Yes No Yes Unknown No Yes Unknown No Yes,57.9±68.9 (58.) 99(9.9) 5(.) 67(.) (67.9) () 6(.6) 8(.7) 96(9.) (6.7) 7.5±5.5 (9.6) 5(9.) (98.8) (.) (85.) 6(.6) 77(.9) 7(69.),76.±689. (5.) 85(.5) (.) 7(8.8) 6(7.) () 8 (.) 6(.8) 7(8.) (8.) 7.±5.6 7(6.8) 7(69.9) 87(98.9) (.) 6(85.) 8(.7) 6(.9) (68.) 997.±6. (7.7) (.6) (.8) (.5) 6(56.5) () 5(8.8) (6.8) (.) 7(.) 8.±5.5 7(.8) 5(.5) 55(98.) (.8) 9(86.) 8(.) 6(7.6) (7.) No 7(7.) (7.9) 7(67.) Homeoxygentherapy Yes 7(9.9) 55(9.) 8 (.7) Unknown 5 No (6.) 99(6.5) (6.) Historyofvaccination Yes 8(8.6) 6(8.5) (8.9) Unknown No (9.5) 7(9.9) 5(89.) Respiratoryphysiotherapy Yes (9.5) 7(9.) 6(.7) Unknown 6 Bodytemperature < 7. >_ 7.< 7.5 >_ 7.5< 9. >_ 9. (7.7) 87(.9) (.6) 7(.8) 8(.7) 65(.) 9(8.) 6(.) 6(7.6) (7.9) 9(.8) (.7) Severe (5.8) 9(7.) (.) Cough Slight 9(5.8) 98(5.) (5.) None 6(.) (.6) (5.) Statistical Test a) Pe=.869 Pe=.8 Pt=.566 Pt=.5 Pe=.599 Pe=. Pt=. Pe=.8 Pe=.78 Pe=.7 Pt=.7 Pe=.56 Pe=.667 Pe=.895 Pe=.67 Pe=.68 Pe=.96 Pe=.798 Pe=.7 Pe=.59 (Continued)
6 Table. (Continued) Variables Total(%) LVFX(%) β -lactam (%) Statistical Test a) Sputum volume Marked Moderate Slight None (.) 5(6.) 66(6.5). (.) (6.9) 8(5.). 9(5.5) (5.) 8(.). Pe=.699 Sputum color Yelow-green Yelow Lightyelow Whiteorclear (8.) 8(5.) 6(.5) (6.) 7(8.9) (5.9) (.) 7(.) (5.) 5(.) 7(9.) (.) Pe=.69 WBC(/mm ) < 8, >_ 8,<, >_,< 5, >_ 5, Notdetermined 98(6.7) 5(.8) 5(5.) 9(.) 9 75(5.7) 7(.6) 6(8.) 6(.7) 7 (5.) (8.) 7(5.) (6.5) Pe=.5 CRP(mg/dL) <.7 >_.7< 5. >_ 5.< >_ Notdetermined (9.5) 9(56.7) 8(8.) (5.7) 9 8(.) 86(5.) (8.) (8.) 7 (.) (7.7) 8(7.) (6.5) Pe=.657 a) Pe:Fisher sexactprobabilitytest,pt:t-test Efficacy rate (%) LVFX group (N9) -lactam group (N58) COX regression analysis: P Fig.. Timecourseoftheeficacyrates. Streptococcus pneumoniaehaemophilus influenzaemoraxella catarrhalis S. pneumoniaeh. influenzaem. catarrhalis S. pneumoniae H. influenzae H. influenzae β Haemophilus influenzae M. catarrhalis S. pneumoniae Mycoplasma pneumoniaechlamydia pneumoniae Mycoplasma pneumoniae
7 ) Sputum volume ) Sputum color 9 LVFX group (N9) -lactam group (N58) 9 LVFX group (N9) -lactam group (N58) 8 8 Improvement rate (%) Improvement rate (%) COX regression analysis: P. COX regression analysis: P ) Cough ) Body temperature 9 8 LVFX group (N88) -lactam group (N55) 9 8 Improvement rate (%) Improvement rate (%) LVFX group (N58) -lactam group (N9) COX regression analysis: P. COX regression analysis: P Fig.. Time course ofimprovementratesforspecific symptoms (sputum volume,sputum color,cough,and body temperature). Chlamydia pneumoniae β β
8 ) 7.5 ) Efficacy rate (%) 6 5 LVFX group (N65) -lactam group (N) Efficacy rate (%) 6 5 LVFX group (N97) -lactam group (N) COX regression analysis: P.96 COX regression analysis: P Fig.. Eficacyratesstratifiedbythebaselinebodytemperature. ) Mildmoderate (FEV % pred 5) ) Severeextremely severe (FEV % pred 5%) 9 LVFX group (N8) -lactam group (N6) 9 LVFX group (N6) -lactam group (N) Efficacy rate (%) 6 5 Efficacy rate (%) 6 5 COX regression analysis: P.6 COX regression analysis: P Fig.5. EficacyratesstratifiedbytheseverityofCOPD. β III β β
9 ) Time course of QOL ) Improvement of QOL (QOL at each assessment-qol on day ) LVFX group (N67) -lactam group (N5) 5 LVFX group (N67) -lactam group (N5) * * * * * ** ** ** ** VAS 5 VAS Wilcoxon rank-sum test : P.5, : P. Repeated measures ANOVA: P. Wilcoxon rank-sum test : P.5, : P. Repeated measures ANOVA: P Fig.6. EvaluationofQOL. ) Mildmoderate (FEV % pred 5%) ) Severeextremely severe (FEV % pred 5%) 5 LVFX group (N7) -lactam group (N) 5 LVFX group (N9) -lactam group (N6) VAS VAS Wilcoxon rank-sum test : P.5 Repeated measures ANOVA: P.76 Wilcoxon rank-sum test : P.5, : P. Repeated measures ANOVA: P Fig.7. ImprovementofQOLstratifiedbytheseverityofCOPD. Table. Readmissionrate No.ofsubjects Group Patientsreadmited(%) Patientsgivenanantibacterialagent atreadmission(%) LVFX 9 (.5) (6.) β -lactam 58 (.) (9.) Reasonforreadmission:Noimprovementofbaselinesymptoms a) Fisher sexactprobabilitytest Statisticaltest a) P=.88 P=.
10 Percentage of patients with isolated causative bacteria Streptococcus pneumoniae 8.8% (7/8).% (/8) Haemophilus influenzae.% (8/8) Moraxella catarrhalis 5.% (/8) Pseudomonas aeruginosa.% (/8) Haemophilus parainfluenzae 8.8% (7/8) Klebsiella pneumoniae.5% (/8) Other bacteria Gram-positive bacteria Gram-negative bacteria.5% (/8).8% (/8) Fig.8. Isolatedcausativebacteriaandpercentageofpatients. Table. MICsofantibacterialagentsforthecausativebacteria )S.pneumoniae Drug <_.6..5 MIC <_ Range MIC5 MIC9 LVFX CFPN-PI CDTR-PI CFDN ABPC CVA/AMPC 5 5 <_.6.5 <_.6.5 <_.6 <_.6 <_ <_.6 <_ )H.influenzae Drug <_.6..5 MIC <_ Range MIC5 MIC9 LVFX CFPN-PI CDTR-PI CFDN ABPC CVA/AMPC 8 5 <_.6 <_.6 <_ <_.5 6 <_ <_ <_ 6 )M.catarhalis Drug <_.6..5 MIC <_ Range MIC5 MIC9 LVFX CFPN-PI CDTR-PI CFDN ABPC CVA/AMPC 8 6 <_.6 <_.6 <_ <_.6.5 <_ <_ β
11 oitacifissal oitacifissal Table5. Causativebacteriadetectedatthetimeofacuteexacerbationandrecurence No. Group Exacerbation LVFX Streptococcuspneumoniae β -lactam Pseudomonasaeruginosa Moraxelacatarhalis β -lactam Haemophilusinfluenzae Haemophilusinfluenzae LVFX Moraxelacatarhalis Streptococcuspneumoniae 5 LVFX Haemophilusinfluenzae Moraxelacatarhalis 6 LVFX Haemophilusinfluenzae 7 LVFX Haemophilusparainfluenzae Streptococcuspyogenes * ThesamegeneticpaternwasshownbyPFGE. Recurence Streptococcuspneumoniae * Notdetected Moraxelacatarhalis Notdetected Haemophilusparainfluenzae Streptococcuspneumoniae Notdetected Table6. Incidenceofadversedrugreactions Group Patientsevaluatedforsafety Patientswithadversedrugreactions Incidenceofadversedrugreactions Skin n C Gastrointestinal CNS Hepatic Renal Others LVFX 96 7.% Nausea(mild:event) Vomiting(mild:event) Dulheadache(mild:event) Hepaticdysfunction(mild:events) Hepaticdysfunction(moderate:event) Renaldysfunction(mild:event) Epistaxis(mild:event) β -lactam 88.5% Redness/pruritus(mild:event) Diarhea(mild:event) Gastritis(mild:event) Table7. Incidenceofadversedrugreactionsinelderlypatients(>_ 75years) Group Patientsevaluatedforsafety Patientswithadversedrugreactions Incidenceofadversedrugreactions Skin n C Gastrointestinal Hepatic Renal LVFX.5% Hepaticdysfunction (mild:event) Renaldysfunction (mild:event) β -lactam 5.8% Redness/pruritus(mild:event) Gastritis(mild:event) S. pneumoniaeh. influenzaem. catarrhalis β S. pneumoniae H. influenzaem. catarrhalis µ H. influenzae
12 β β β β S. pneumoniaeh. influenzaem. catarrhalis Mycoplasma pneumoniaechlamydia pneumoniae Chlamydia pneumoniae Mycoplasma pneumoniaechlamydia pneumoniae
13
14 Mycoplasma pneumoniae Mycoplasma pneumoniae
15 Chlamydia pneumoniae Chlamydia pneumoniae Chlamydia pneumoniae Chlamydia pneumoniae Chlamydophila pneumoniae
16 β β β β β β β β Streptococcus pneumoniae Haemophilus influenzae Moraxella catarrhalis Mycoplasma pneumoniae Chlamydia pneumoniae β β
日本化学療法学会雑誌第59巻第5号
 Streptococcus pneumoniae Haemophilus influenzae Moraxella catarrhalis S. pneumoniae H. influenzae M. catarrhalis S. pneumoniae H. influenzae M. catarrhalis S. pneumoniae H. influenzae M. catarrhalis S.
Streptococcus pneumoniae Haemophilus influenzae Moraxella catarrhalis S. pneumoniae H. influenzae M. catarrhalis S. pneumoniae H. influenzae M. catarrhalis S. pneumoniae H. influenzae M. catarrhalis S.
日本化学療法学会雑誌第58巻第4号
 Escherichia coli Enterococcus faecalisstreptococcus agalactiae Klebsiella pneumoniae Staphylococcus epidermidis E. colie. faecalispseudomonas aeruginosa K. pneumoniae S. agalactiae E. coli E. coli μ p
Escherichia coli Enterococcus faecalisstreptococcus agalactiae Klebsiella pneumoniae Staphylococcus epidermidis E. colie. faecalispseudomonas aeruginosa K. pneumoniae S. agalactiae E. coli E. coli μ p
日本化学療法学会雑誌第60巻第4号
 Streptococcus pneumoniae Haemophilus influenzae S. pneumoniae H. influenzae Key words β Streptococcus pneumoniae Haemophilus influenzae S. pneumoniae H. influenzae μ μ S. pneumoniae H. influenzae S. pneumoniae
Streptococcus pneumoniae Haemophilus influenzae S. pneumoniae H. influenzae Key words β Streptococcus pneumoniae Haemophilus influenzae S. pneumoniae H. influenzae μ μ S. pneumoniae H. influenzae S. pneumoniae
日本化学療法学会雑誌第57巻第4号
 Streptococcus pneumoniae Haemophilus influenzae β β Key words I β Enterococcus faecium Pseudomonas aeruginosa Streptococcus pneumoniae S. pneumoniae Haemophilus influenzae S. pneumoniae H. influenzae Table.
Streptococcus pneumoniae Haemophilus influenzae β β Key words I β Enterococcus faecium Pseudomonas aeruginosa Streptococcus pneumoniae S. pneumoniae Haemophilus influenzae S. pneumoniae H. influenzae Table.
日本化学療法学会雑誌第61巻第6号
 β Moraxella catarrhalis Escherichia coli Citrobacter Klebsiella pneumoniae Enterobacter cloacae Serratia marcescens Proteus Pseudomonas aeruginosa Acinetobacter Bacteroides fragilis β Haemophilus influenzae
β Moraxella catarrhalis Escherichia coli Citrobacter Klebsiella pneumoniae Enterobacter cloacae Serratia marcescens Proteus Pseudomonas aeruginosa Acinetobacter Bacteroides fragilis β Haemophilus influenzae
Table 1. Antibacterial activitiy of grepafloxacin and other antibiotics against clinical isolates
 Table 1. Antibacterial activitiy of grepafloxacin and other antibiotics against clinical isolates Table 2-1. Summary of patients treated with grepafloxacin for respiratory infection 1) Out: outpatient,
Table 1. Antibacterial activitiy of grepafloxacin and other antibiotics against clinical isolates Table 2-1. Summary of patients treated with grepafloxacin for respiratory infection 1) Out: outpatient,
日本化学療法学会雑誌第56巻第3号
 β Key words Candida albicans albicans Candida C. albicans Candida glabrata Candida krusei albicans Candida C. glabrata C. albicans albicans Candida β in vitro in vivo C. glabrata C. krusei I β γ µ Candida
β Key words Candida albicans albicans Candida C. albicans Candida glabrata Candida krusei albicans Candida C. glabrata C. albicans albicans Candida β in vitro in vivo C. glabrata C. krusei I β γ µ Candida
CHEMOTHERAPY FEB Table 1. Activity of cefpirome and others against clinical isolates
 VOL.39 S-1 CHEMOTHERAPY FEB. 1981 Table 1. Activity of cefpirome and others against clinical isolates VOL.39 S-1 CHEMOTHERAPY FEB. 1991 72 M, 55.5 kg 66 F, 53 kg Chronic bronchitis Bronchopneumonia Peak
VOL.39 S-1 CHEMOTHERAPY FEB. 1981 Table 1. Activity of cefpirome and others against clinical isolates VOL.39 S-1 CHEMOTHERAPY FEB. 1991 72 M, 55.5 kg 66 F, 53 kg Chronic bronchitis Bronchopneumonia Peak
日本化学療法学会雑誌第57巻第1号
 In vitro Streptococcus pneumoniae Escherichia coli in vitro Streptococcus pneumoniae Escherichia coli µs. pneumoniae E. coli µ Key words in vitrostreptococcus pneumoniae Escherichia coli Escherichia coli
In vitro Streptococcus pneumoniae Escherichia coli in vitro Streptococcus pneumoniae Escherichia coli µs. pneumoniae E. coli µ Key words in vitrostreptococcus pneumoniae Escherichia coli Escherichia coli
日本化学療法学会雑誌第58巻第2号
 Key words β Candida Table1. MCFGCPAstudygroup Investigator (representative) YoshitsuguMiyazaki NaoyukiMiyashita RyoichiAmitani KenjiOgawa AtsuyukiKurashima ToshiroKiguchi MichiakiMishima YuichiInoue HiroshiSaito
Key words β Candida Table1. MCFGCPAstudygroup Investigator (representative) YoshitsuguMiyazaki NaoyukiMiyashita RyoichiAmitani KenjiOgawa AtsuyukiKurashima ToshiroKiguchi MichiakiMishima YuichiInoue HiroshiSaito
CHEMOTHERAPY
 CHEMOTHERAPY VOL.41 S-2 Laboratory and clinical evaluation of teicoplanin CHEMOTHERAPY AUG. 1993 VOL.41 S-2 Laboratory and clinical evaluation of teicoplanin Table 1. Comparative in vitro activity of teicoplanin
CHEMOTHERAPY VOL.41 S-2 Laboratory and clinical evaluation of teicoplanin CHEMOTHERAPY AUG. 1993 VOL.41 S-2 Laboratory and clinical evaluation of teicoplanin Table 1. Comparative in vitro activity of teicoplanin
日本化学療法学会雑誌第57巻第S-2号
 Key words β I Table. Observationandtestschedule Testschedule Observation/Tests Beforetreatment Endoftreatment 5 9days aftercompletion oftreatment 4 6weeks aftercompletion oftreatment Informedconsent Patientbackground
Key words β I Table. Observationandtestschedule Testschedule Observation/Tests Beforetreatment Endoftreatment 5 9days aftercompletion oftreatment 4 6weeks aftercompletion oftreatment Informedconsent Patientbackground
THE JAPANESE JOURNAL OF ANTIBIOTICS 68 3 June 2015 Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis % 2 S. pneumon
 June 2015 THE JAPANESE JOURNAL OF ANTIBIOTICS 68 3 189 49 1 : 14 1 2 2 3 1 2 3 2015 4 3 1 : 14 CVA/AMPC 1 : 14 27 CVA/AMPC 1 : 14 88.5% Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis
June 2015 THE JAPANESE JOURNAL OF ANTIBIOTICS 68 3 189 49 1 : 14 1 2 2 3 1 2 3 2015 4 3 1 : 14 CVA/AMPC 1 : 14 27 CVA/AMPC 1 : 14 88.5% Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis
Fig.2. Sensitivity distribution of clinical isolates of S. epidermidis (24 strains, 106 CFU/ml) Staphylococcus aureus Staphylococcus epider- midis Ent
 Fig.2. Sensitivity distribution of clinical isolates of S. epidermidis (24 strains, 106 CFU/ml) Staphylococcus aureus Staphylococcus epider- midis Enterococcus faecalis Klebsiella pneumoniae, Morganella
Fig.2. Sensitivity distribution of clinical isolates of S. epidermidis (24 strains, 106 CFU/ml) Staphylococcus aureus Staphylococcus epider- midis Enterococcus faecalis Klebsiella pneumoniae, Morganella
366 12 THE JAPANESE JOURNAL OF ANTIBIOTICS 65 6 Dec. 2012 1 8 DNA 2,3 16 12 20 171 2008 12 2010 11 2 3,558 4.44% 1.65% 1.17% 90% 9 Escherichia coli -
 Dec. 2012 THE JAPANESE JOURNAL OF ANTIBIOTICS 65 6 365 11 sita oxacin 1 1 1 1 1 1 2 2 3 3 1 1 1 2 3 2012 9 14 sita oxacin STFX 50 mg 10% 2008 1 2008 12 2010 11 2 STFX 1,452 91.4% 1,235/1,351 95.9% 466/486
Dec. 2012 THE JAPANESE JOURNAL OF ANTIBIOTICS 65 6 365 11 sita oxacin 1 1 1 1 1 1 2 2 3 3 1 1 1 2 3 2012 9 14 sita oxacin STFX 50 mg 10% 2008 1 2008 12 2010 11 2 STFX 1,452 91.4% 1,235/1,351 95.9% 466/486
VOL.42 S-1
 CHEMOTHERAPY APR. 1994 VOL.42 S-1 CHEMOTHERAPY APR. 1994 Table 1. Criteria for evaluation of clinical efficacy by the Japanese Society of Oral and Maxillo-Facial Surgeons Grades of symptoms and numerical
CHEMOTHERAPY APR. 1994 VOL.42 S-1 CHEMOTHERAPY APR. 1994 Table 1. Criteria for evaluation of clinical efficacy by the Japanese Society of Oral and Maxillo-Facial Surgeons Grades of symptoms and numerical
CHEMOTHERAPY Table 1 Clinical effect of Sultamicillin
 CHEMOTHERAPY CHEMOTHERAPY Table 1 Clinical effect of Sultamicillin CHEMOTHERAPY Fig. 1 MICs of sultamicillin against respiratory pathogenic Branhamella catarrhalis 62 strains, inoculum size 106CFU/m1 Fig.
CHEMOTHERAPY CHEMOTHERAPY Table 1 Clinical effect of Sultamicillin CHEMOTHERAPY Fig. 1 MICs of sultamicillin against respiratory pathogenic Branhamella catarrhalis 62 strains, inoculum size 106CFU/m1 Fig.
CHEMOTHERAPY JUNE 1993 Table 1. Background of patients in pharmacokinetic study
 CHEMOTHERAPY JUNE 1993 Table 1. Background of patients in pharmacokinetic study VOL. 41 S 1 Table 2. Levels (Đg/ml or Đg/g) of S-1006 in serum, bile, and tissue (gallbladder) after oral administration
CHEMOTHERAPY JUNE 1993 Table 1. Background of patients in pharmacokinetic study VOL. 41 S 1 Table 2. Levels (Đg/ml or Đg/g) of S-1006 in serum, bile, and tissue (gallbladder) after oral administration
日本化学療法学会雑誌第54巻第S-1号
 β β β Key words β β I HP- -CD CHR RHC R R R R CHR R R R R RHC R R R CHR R R R RHC CHR R H CH CH Fig.. Structuralformulaofhydroxypropyl-β -cyclodextrin(hp-β -CD). CH n H β β β β Table-. Dosageandadministration(Singleadministrationstudy)
β β β Key words β β I HP- -CD CHR RHC R R R R CHR R R R R RHC R R R CHR R R R RHC CHR R H CH CH Fig.. Structuralformulaofhydroxypropyl-β -cyclodextrin(hp-β -CD). CH n H β β β β Table-. Dosageandadministration(Singleadministrationstudy)
CHEMOTHERAPY
 CHEMOTHERAPY CHEMOTHERAPY Table 1 Antibacterial activity of Sulbactam/CPZ against standard strains MIC mg/ml Inoculum size 106 CFU/ml * Sulbactam/CPZ= 1: 1 ** Concentration of Sulbactam+ CPZ CHEMOTHERAPY
CHEMOTHERAPY CHEMOTHERAPY Table 1 Antibacterial activity of Sulbactam/CPZ against standard strains MIC mg/ml Inoculum size 106 CFU/ml * Sulbactam/CPZ= 1: 1 ** Concentration of Sulbactam+ CPZ CHEMOTHERAPY
untitled
 1988 2000 2002 2004 2006 2008 IFN Lamivudine Adefovir Entecavir 1 Total number 560 Sex (male/female) 424/136 Age (years)* 38 (15-68) Duration of treatment (weeks)* 26 (1-592) Follow-up time (years) 75(05-21
1988 2000 2002 2004 2006 2008 IFN Lamivudine Adefovir Entecavir 1 Total number 560 Sex (male/female) 424/136 Age (years)* 38 (15-68) Duration of treatment (weeks)* 26 (1-592) Follow-up time (years) 75(05-21
日本化学療法学会雑誌第65巻第3号
 μ Key words Chlamydia trachomatis C. trachomatis I Table1.Observation items, categories and scores Observation item Category Score Body temperature () Lower abdominal pain Uterine corpus tenderness Condition
μ Key words Chlamydia trachomatis C. trachomatis I Table1.Observation items, categories and scores Observation item Category Score Body temperature () Lower abdominal pain Uterine corpus tenderness Condition
epidermidis, Enterococcus faecalis, Enterococcus Klebsiella pneumoniae, Proteus mirabilis, indolepositive Proteus spp., Enterobacter spp., Serratia
 epidermidis, Enterococcus faecalis, Enterococcus Klebsiella pneumoniae, Proteus mirabilis, indolepositive Proteus spp., Enterobacter spp., Serratia Table 3. Overall clinical efficacy of cefozopran in
epidermidis, Enterococcus faecalis, Enterococcus Klebsiella pneumoniae, Proteus mirabilis, indolepositive Proteus spp., Enterobacter spp., Serratia Table 3. Overall clinical efficacy of cefozopran in
208 ( 2 ) THE JAPANESE JOURNAL OF ANTIBIOTICS 63 _ 3 June 2010 Cefditoren pivoxil (CDTR-PI) MS MS 10%
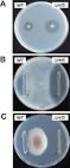 207 ( 1 ) Cefditoren pivoxil G 2010 2 2 2006 3 Cefditoren pivoxil CDTR-PI MS 10% CDTR-PI 305 2,144 2,006 1,958 1.79% 36 2,006 26 (1.30%) CDTR-PI CDTR-PI (9 mg/kg/day) 1.5 2 (2.70%) 2 (1.92%) 93.5% 1,831
207 ( 1 ) Cefditoren pivoxil G 2010 2 2 2006 3 Cefditoren pivoxil CDTR-PI MS 10% CDTR-PI 305 2,144 2,006 1,958 1.79% 36 2,006 26 (1.30%) CDTR-PI CDTR-PI (9 mg/kg/day) 1.5 2 (2.70%) 2 (1.92%) 93.5% 1,831
Fig. 1 Chemical structure of DL-8280
 Fig. 1 Chemical structure of DL-8280 Fig. 2 Susceptibility of cl in ical isolates to DL4280 Fig. 5 Susceptibility of clinical isolates to DL-8280 Fig. 3 Susceptibility of clinical isolates to DL-8280 Fig.
Fig. 1 Chemical structure of DL-8280 Fig. 2 Susceptibility of cl in ical isolates to DL4280 Fig. 5 Susceptibility of clinical isolates to DL-8280 Fig. 3 Susceptibility of clinical isolates to DL-8280 Fig.
CHEMOTHERAPY
 CHEMOTHERAPY CHEMOTHERAPY Table 1 Antibacterial activity of BRL 28500 against standard strains of bacteria Fig, 1 Sensitivity distribution of ABPC-resistant E. coli isolated from urinary tract Fig. 2 Sensitivity
CHEMOTHERAPY CHEMOTHERAPY Table 1 Antibacterial activity of BRL 28500 against standard strains of bacteria Fig, 1 Sensitivity distribution of ABPC-resistant E. coli isolated from urinary tract Fig. 2 Sensitivity
Table 1. Antibacterial spectrum SBT ABPC ABPC CPZ : sulbactamiampicillin : ampicillin : cefoperazone
 Table 1. Antibacterial spectrum SBT ABPC ABPC CPZ : sulbactamiampicillin : ampicillin : cefoperazone (inoculum size= 106 CFU/ml) (Ĉ-lactamase producer : 2 strains) Fig. 1. Sensitivity distribution of
Table 1. Antibacterial spectrum SBT ABPC ABPC CPZ : sulbactamiampicillin : ampicillin : cefoperazone (inoculum size= 106 CFU/ml) (Ĉ-lactamase producer : 2 strains) Fig. 1. Sensitivity distribution of
日本化学療法学会雑誌第66巻第2号
 173 Table 1.Detection rates of Neisseria gonorrhoeae, Chlamydia trachomatis, Mycoplasma genitalium, Ureaplasma urealyticum, Haemophilus influenzae and adenovirus from male urethritis in Urethritis No.
173 Table 1.Detection rates of Neisseria gonorrhoeae, Chlamydia trachomatis, Mycoplasma genitalium, Ureaplasma urealyticum, Haemophilus influenzae and adenovirus from male urethritis in Urethritis No.
日本化学療法学会雑誌第51巻第2号
 piperacillin piperacillin PIPC. g Cmax CL PIPC CL CLR CLNR CL PIPC g g Cmax PIPC Key words: piperacillin Piperacillin PIPC PIPC g g PIPC Cmax g g ml g g ml g g ml T T T PIPC g g T Ccr ml min AUCCmax PIPC
piperacillin piperacillin PIPC. g Cmax CL PIPC CL CLR CLNR CL PIPC g g Cmax PIPC Key words: piperacillin Piperacillin PIPC PIPC g g PIPC Cmax g g ml g g ml g g ml T T T PIPC g g T Ccr ml min AUCCmax PIPC
VOL.35 S-2 CHEMOTHERAPY Table 1 Sex and age distribution Table 2 Applications of treatment with carumonam Table 3 Concentration of carumonam in human
 CHEMOTHERAPY Fig. 1 Chemical structure of carumonam Disodium(+)-(Z)-CCE1-(2-amino-4-thiazoly1)-2-[[(2S, -(carbamoyloxymethyl)-4-oxo-1-sulfonato-3-azetidinyll -2-oxoethylidene] amino] oxy] acetate 3S)-2
CHEMOTHERAPY Fig. 1 Chemical structure of carumonam Disodium(+)-(Z)-CCE1-(2-amino-4-thiazoly1)-2-[[(2S, -(carbamoyloxymethyl)-4-oxo-1-sulfonato-3-azetidinyll -2-oxoethylidene] amino] oxy] acetate 3S)-2
CHEMOTHERAPY JUNE 1986
 VOL. 34 S-3 CHEMOTHERAPY Fig. 1 Structural formula of L-105 CHEMOTHERAPY JUNE 1986 VOL. 34 S-3 CHEMOTHERAPY Table 1 Antibacterial spectra of L-105 against gram negative anaerobic rods Inoculum 106 cells/ml
VOL. 34 S-3 CHEMOTHERAPY Fig. 1 Structural formula of L-105 CHEMOTHERAPY JUNE 1986 VOL. 34 S-3 CHEMOTHERAPY Table 1 Antibacterial spectra of L-105 against gram negative anaerobic rods Inoculum 106 cells/ml
VOL.39 S-3
 VOL.39 S-3 CHEMOTHERAPY SEPT.1991 Table 1. Background of characteristics and allocation of 5 healthy male volunteers in a multiple-dose study on panipenem/betamipron Day 1 Fig. 1. Schedule of multiple-dose
VOL.39 S-3 CHEMOTHERAPY SEPT.1991 Table 1. Background of characteristics and allocation of 5 healthy male volunteers in a multiple-dose study on panipenem/betamipron Day 1 Fig. 1. Schedule of multiple-dose
Fig. 1 Chemical structure of norfioxacin (AM-715)
 Fig. 1 Chemical structure of norfioxacin (AM-715) Table 1 Serum and biliary concentration of norfloxacin (AM-715) Table 2 Protocol for clinical evaluation of norfloxacin (AM-715) in the treatment of biliary
Fig. 1 Chemical structure of norfioxacin (AM-715) Table 1 Serum and biliary concentration of norfloxacin (AM-715) Table 2 Protocol for clinical evaluation of norfloxacin (AM-715) in the treatment of biliary
THE JAPANESE JOURNAL OF ANTIBIOTICS 67 3 June 2014 STFX mg mg mg mg mg 1
 June 2014 THE JAPANESE JOURNAL OF ANTIBIOTICS 67 3 175 29 1 2 2 2 2 2 3 4 4 5 5 2 2 1 2 3 4 5 2014 4 15 STFX 50 mg 10% 2011 8 1 100 mg 1 1 2011 12 2013 5 226 1,186 1,089 1,069 2.11% 23/1,089 1.10% 12/1,089
June 2014 THE JAPANESE JOURNAL OF ANTIBIOTICS 67 3 175 29 1 2 2 2 2 2 3 4 4 5 5 2 2 1 2 3 4 5 2014 4 15 STFX 50 mg 10% 2011 8 1 100 mg 1 1 2011 12 2013 5 226 1,186 1,089 1,069 2.11% 23/1,089 1.10% 12/1,089
MIC MIC...
 50 mg 10% 2.7.36 2.7.36 2.7.36... 1 1.6... 1 2.6... 3 3.6... 5 3.16... 5 3.26... 12 3.36... 16 4.6... 17 5.6... 19 6.6... 20 2.7.3.3.16-1 MIC... 9 2.7.3.3.16-2 MIC... 10 2.7.3.3.16-3 MIC E. coli... 11
50 mg 10% 2.7.36 2.7.36 2.7.36... 1 1.6... 1 2.6... 3 3.6... 5 3.16... 5 3.26... 12 3.36... 16 4.6... 17 5.6... 19 6.6... 20 2.7.3.3.16-1 MIC... 9 2.7.3.3.16-2 MIC... 10 2.7.3.3.16-3 MIC E. coli... 11
日本化学療法学会雑誌第50巻第6号
 13 11 30 14 4 22 Streptococcus pneumoniae Haemophilus influenzae amoxicillin AMPC cefditoren pivoxil CDTR AMPC 50 mg kg day CDTR 9mg kg day CDTR 18 mg kg day S. pneumoniae H. influenzae 8 17 25 1 25 S.
13 11 30 14 4 22 Streptococcus pneumoniae Haemophilus influenzae amoxicillin AMPC cefditoren pivoxil CDTR AMPC 50 mg kg day CDTR 9mg kg day CDTR 18 mg kg day S. pneumoniae H. influenzae 8 17 25 1 25 S.
日本消化器外科学会雑誌第30巻第3号
 Key words : gastric cancer, progltostic factor. multivariate analysis Factors selected after Coffelation- after Stepwise rnitrally check check H o s t l A g e C O 2 S e x C O 3 Pre-op. compliation O Treatment
Key words : gastric cancer, progltostic factor. multivariate analysis Factors selected after Coffelation- after Stepwise rnitrally check check H o s t l A g e C O 2 S e x C O 3 Pre-op. compliation O Treatment
Clostridium difficile ciprofloxacin, ofloxacin, norfloxacin Bifidobacterium Lactobacillus Lactobacillus Bacteroides fragilis B. fragilis C. difficile
 Clostridium difficile ciprofloxacin, ofloxacin, norfloxacin Bifidobacterium Lactobacillus Lactobacillus Bacteroides fragilis B. fragilis C. difficile Key words: temafloxacin, TA-167, Bacteroides fragilis,
Clostridium difficile ciprofloxacin, ofloxacin, norfloxacin Bifidobacterium Lactobacillus Lactobacillus Bacteroides fragilis B. fragilis C. difficile Key words: temafloxacin, TA-167, Bacteroides fragilis,
R06_01
 Staphylococcus aureus (MSSA) PCG (N=118,334) 57,369 (48.5%) 判定不能 :3 (0.0%) 60,962 (51.5%) CEZ (N=143,723) I:42 (0.0%) 143,635 (99.9%) R:46 (0.0%) CVA/AMPC (N=19,281) R:14 (0.1%) 19,265 (99.9%) 判定不能 :2
Staphylococcus aureus (MSSA) PCG (N=118,334) 57,369 (48.5%) 判定不能 :3 (0.0%) 60,962 (51.5%) CEZ (N=143,723) I:42 (0.0%) 143,635 (99.9%) R:46 (0.0%) CVA/AMPC (N=19,281) R:14 (0.1%) 19,265 (99.9%) 判定不能 :2
 CHEMOTHERAPY Table 2 Clinical response of 6059-S by infection Table 3 Effect of 6059-S on blood chemistry *Normal range : S-GOT 20 `60 mu/ml, S-GPT 5 `25 IU/L, Al-Pase 30 `85 mu/ml In oilier cases : S-GOT
CHEMOTHERAPY Table 2 Clinical response of 6059-S by infection Table 3 Effect of 6059-S on blood chemistry *Normal range : S-GOT 20 `60 mu/ml, S-GPT 5 `25 IU/L, Al-Pase 30 `85 mu/ml In oilier cases : S-GOT
400 46 THE JAPANESE JOURNAL OF ANTIBIOTICS 65 6 Dec. 2012 LVFX 100 mg 3 / 7 150 mg 2 / 7 2 2006 2008 9 LVFX PK PD 2009 7 100 mg 1 3 500 mg 1 1 AUC/MIC
 Dec. 2012 THE JAPANESE JOURNAL OF ANTIBIOTICS 65 6 399 45 2012 11 5 LVFX 500 mg 1 1 20 Chlamydia trachomatis C. trachomatismycoplasma genitalium M. genitalium LVFX 1 500 mg 1 1 7 22 22 C. trachomatis 17
Dec. 2012 THE JAPANESE JOURNAL OF ANTIBIOTICS 65 6 399 45 2012 11 5 LVFX 500 mg 1 1 20 Chlamydia trachomatis C. trachomatismycoplasma genitalium M. genitalium LVFX 1 500 mg 1 1 7 22 22 C. trachomatis 17
 The clinical characteristics of Mycoplasma pneumoniae pneumonia in children younger than 6 years old Nobue Takeda1,2),Tomomichi Kurosaki1),Naruhiko Ishiwada2),Yoichi Kohno2) 1)Department of Pediatrics,Chiba
The clinical characteristics of Mycoplasma pneumoniae pneumonia in children younger than 6 years old Nobue Takeda1,2),Tomomichi Kurosaki1),Naruhiko Ishiwada2),Yoichi Kohno2) 1)Department of Pediatrics,Chiba
 Table 1. Antibacterial activity of cefdinir, cefixime, cefteram, cefuroxime, cefaclor and amoxicillin against standard strains Inoculum size: 108 cells/ml CFDN: cefdinir, CFIX: cefixime, CFTM: cefteram,
Table 1. Antibacterial activity of cefdinir, cefixime, cefteram, cefuroxime, cefaclor and amoxicillin against standard strains Inoculum size: 108 cells/ml CFDN: cefdinir, CFIX: cefixime, CFTM: cefteram,
食道がん化学放射線療法後のsalvage手術
 2006 2 17 52 Daly JM, et al. J Am Coll Surg 2000;190:562-573 Esophageal Cancer: Results of an American College of Surgeons Patient Care Evaluation Study Daly JM, et al. J Am Coll Surg 2000;190:562-573
2006 2 17 52 Daly JM, et al. J Am Coll Surg 2000;190:562-573 Esophageal Cancer: Results of an American College of Surgeons Patient Care Evaluation Study Daly JM, et al. J Am Coll Surg 2000;190:562-573
一般社団法人 日本感染症学会 第89回総会資料
 Longitudinal surveillance of Haemophilus influenzae isolates from pediatric patients with meningitis throughout Japan, 2000 2011 Journal of Infection and Chemotherapy Vol.19No.1 p34-41kimiko Ubukata Long-term
Longitudinal surveillance of Haemophilus influenzae isolates from pediatric patients with meningitis throughout Japan, 2000 2011 Journal of Infection and Chemotherapy Vol.19No.1 p34-41kimiko Ubukata Long-term
日本化学療法学会雑誌第61巻第4号
 μ μ μ μ μ μ Key words I β μ Sex Age (years) Height (cm) Evaluation items Table1.Characteristics of patients 1. mg/kg/day (n14) 2.5 mg/kg/day (n9) 5. mg/kg/day (n9) Total (n32) male 12 8 7 27 female 2 1
μ μ μ μ μ μ Key words I β μ Sex Age (years) Height (cm) Evaluation items Table1.Characteristics of patients 1. mg/kg/day (n14) 2.5 mg/kg/day (n9) 5. mg/kg/day (n9) Total (n32) male 12 8 7 27 female 2 1
Table1MIC of BAY o 9867 against standard strains
 Table1MIC of BAY o 9867 against standard strains Fig.2Cumulative and Distribution Curves of MIC (S.aureus 54 strains) 106cfu/ml Fig.3Correlogram of MIC (S.aureus 54 strains) CHEMOTHERAPY 451 Fig.4Cumulative
Table1MIC of BAY o 9867 against standard strains Fig.2Cumulative and Distribution Curves of MIC (S.aureus 54 strains) 106cfu/ml Fig.3Correlogram of MIC (S.aureus 54 strains) CHEMOTHERAPY 451 Fig.4Cumulative
 Osamu NEMOTO, M.D. Clinical and Bacteriological Research of Sucrose/ Povidone-Iodine Ointment (U-PASTA kowa) for Pressure Sores and Skin Ulcers Osamu Nemoto Department of Dermatology,Tonan Hosptal
Osamu NEMOTO, M.D. Clinical and Bacteriological Research of Sucrose/ Povidone-Iodine Ointment (U-PASTA kowa) for Pressure Sores and Skin Ulcers Osamu Nemoto Department of Dermatology,Tonan Hosptal
4月号 学会特集号 122247/16)一般演題目次
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 48 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 58 59 49 60 61 62 63 64 65 66 67 68
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 48 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 58 59 49 60 61 62 63 64 65 66 67 68
Key words: bacterial meningitis, Haemophilus influenzae type b, Streptococcus pneumoniae, rapid diagnosis, childhood
 Key words: bacterial meningitis, Haemophilus influenzae type b, Streptococcus pneumoniae, rapid diagnosis, childhood Fig.1 Distribution of the cases with bacterial meningitis by age and pathogens Chiba
Key words: bacterial meningitis, Haemophilus influenzae type b, Streptococcus pneumoniae, rapid diagnosis, childhood Fig.1 Distribution of the cases with bacterial meningitis by age and pathogens Chiba
CHEMOTHERAPY JUNE 1987 Table1 Media used *BHIB, brain heart infusion broth (Difco); /3 -NAD, S -nicotinamidoadeninedinucleotide (Sigma Chemical Co.);
 VOL.35 S-2 CHEMOTHERAPY Fig.1 Chemical structure of carumonam CHEMOTHERAPY JUNE 1987 Table1 Media used *BHIB, brain heart infusion broth (Difco); /3 -NAD, S -nicotinamidoadeninedinucleotide (Sigma Chemical
VOL.35 S-2 CHEMOTHERAPY Fig.1 Chemical structure of carumonam CHEMOTHERAPY JUNE 1987 Table1 Media used *BHIB, brain heart infusion broth (Difco); /3 -NAD, S -nicotinamidoadeninedinucleotide (Sigma Chemical
Title 泌尿器科領域に於ける17-Ketosteroidの研究 17-Ketosteroidの臨床的研究 第 III 篇 : 尿 Author(s) 卜部, 敏入 Citation 泌尿器科紀要 (1958), 4(1): 3-31 Issue Date URL
 Title 泌尿器科領域に於ける17-Ketosteroidの研究 17-Ketosteroidの臨床的研究 第 III 篇 : 尿 Author(s) 卜部, 敏入 Citation 泌尿器科紀要 (1958), 4(1): 3-31 Issue Date 1958-01 URL http://hdl.handle.net/2433/111559 Right Type Departmental Bulletin
Title 泌尿器科領域に於ける17-Ketosteroidの研究 17-Ketosteroidの臨床的研究 第 III 篇 : 尿 Author(s) 卜部, 敏入 Citation 泌尿器科紀要 (1958), 4(1): 3-31 Issue Date 1958-01 URL http://hdl.handle.net/2433/111559 Right Type Departmental Bulletin
日本化学療法学会雑誌第57巻第S-2号
 µ µ Key words µ µ µ I Table1. Subjectprofilesin(A)singleand(B)multiple-dosestudies (A)Single-dosestudy Age (yr) Height (cm) Bodyweight (kg) BMI (kg/m ) Ccr(Cockcroft) (ml/min) Ethnicity dose(mg) n 5 5.1±.,3
µ µ Key words µ µ µ I Table1. Subjectprofilesin(A)singleand(B)multiple-dosestudies (A)Single-dosestudy Age (yr) Height (cm) Bodyweight (kg) BMI (kg/m ) Ccr(Cockcroft) (ml/min) Ethnicity dose(mg) n 5 5.1±.,3
 Key words : R-plasmid, Urinary tract infection, E. coli Fig. 1. MIC distribution against E. coli isolated from urinary tract (366 strains) and isolation - frequencies of drug-resistant strains Table 1.
Key words : R-plasmid, Urinary tract infection, E. coli Fig. 1. MIC distribution against E. coli isolated from urinary tract (366 strains) and isolation - frequencies of drug-resistant strains Table 1.
untitled
 19 CSPOR CRC/2009.8.8-9 2009 8 8 ( ) - CSPOR CRC SEMINAR- [email protected] http://www.oncoloplan.com - Toru Watanabe MD - 2 Phase I: Dose Finding trial Phase II : Efficacy and Safety trial Phase
19 CSPOR CRC/2009.8.8-9 2009 8 8 ( ) - CSPOR CRC SEMINAR- [email protected] http://www.oncoloplan.com - Toru Watanabe MD - 2 Phase I: Dose Finding trial Phase II : Efficacy and Safety trial Phase
日本化学療法学会雑誌第53巻第S-3号
 moxifloxacin in vitro moxifloxacin in vitro 17 9 6 17 11 21 moxifloxacinmflx in vitro cefdinir CFDNclavulanic acidamoxicillincvaampcclarithromycincamclindamycincldm levofloxacinlvfx 1MFLX Clostridium clostridiiformeclostridium
moxifloxacin in vitro moxifloxacin in vitro 17 9 6 17 11 21 moxifloxacinmflx in vitro cefdinir CFDNclavulanic acidamoxicillincvaampcclarithromycincamclindamycincldm levofloxacinlvfx 1MFLX Clostridium clostridiiformeclostridium
 Table 1.Quality control of MICs for reference strains Table 2.Antimicrobial activity of gatifloxacin against aerobic bacteria Table 4.Antimicrobial activity of gatifloxacin and other quinolones against
Table 1.Quality control of MICs for reference strains Table 2.Antimicrobial activity of gatifloxacin against aerobic bacteria Table 4.Antimicrobial activity of gatifloxacin and other quinolones against
第65回日本化学療法学会東日本支部総会 抄録
 鍵 76 Clostridioides difficile C. difficile 77 γ γ Mycobacterium avium 78 79 Clostridium difficile Treponema pallidum 80 81 82 in vitro in vitro 83 鍵 Treponema pallidum 84 Chlamydia trachomatis Mycoplasma
鍵 76 Clostridioides difficile C. difficile 77 γ γ Mycobacterium avium 78 79 Clostridium difficile Treponema pallidum 80 81 82 in vitro in vitro 83 鍵 Treponema pallidum 84 Chlamydia trachomatis Mycoplasma
CHEMOTHERAPY JUN Citrobacter freundii 27, Enterobacter aerogenes 26, Enterobacter cloacae 27, Proteus rettgeri 7, Proteus inconstans 20, Proteus
 VOL. 32 S-4 CHEMOTHERAPY Fig. 1 Chemical structure of sodium cefoperazone Fig. 2 Chemical structure of sodium cefoperazone CHEMOTHERAPY JUN. 1984 Citrobacter freundii 27, Enterobacter aerogenes 26, Enterobacter
VOL. 32 S-4 CHEMOTHERAPY Fig. 1 Chemical structure of sodium cefoperazone Fig. 2 Chemical structure of sodium cefoperazone CHEMOTHERAPY JUN. 1984 Citrobacter freundii 27, Enterobacter aerogenes 26, Enterobacter
A Nutritional Study of Anemia in Pregnancy Hematologic Characteristics in Pregnancy (Part 1) Keizo Shiraki, Fumiko Hisaoka Department of Nutrition, Sc
 A Nutritional Study of Anemia in Pregnancy Hematologic Characteristics in Pregnancy (Part 1) Keizo Shiraki, Fumiko Hisaoka Department of Nutrition, School of Medicine, Tokushima University, Tokushima Fetal
A Nutritional Study of Anemia in Pregnancy Hematologic Characteristics in Pregnancy (Part 1) Keizo Shiraki, Fumiko Hisaoka Department of Nutrition, School of Medicine, Tokushima University, Tokushima Fetal
21 Effects of background stimuli by changing speed color matching color stimulus
 21 Effects of background stimuli by changing speed color matching color stimulus 1100274 2010 3 1 ,.,,.,.,.,,,,.,, ( FL10N-EDL). ( 10cm, 2cm),,, 3.,,,, 4., ( MSS206-402W2J), ( SDM496)., 1200r/min,1200r/min
21 Effects of background stimuli by changing speed color matching color stimulus 1100274 2010 3 1 ,.,,.,.,.,,,,.,, ( FL10N-EDL). ( 10cm, 2cm),,, 3.,,,, 4., ( MSS206-402W2J), ( SDM496)., 1200r/min,1200r/min
 Survey of the Incidence of Dry Cough Caused by ACE Inhibitors MOTOHIRO YAGI*, MIKIO SAITO, MANABU ABE and KATSUJI UNO Pharmacy, Suibarago Hospital õ Received October 24, 1996 Accepted September 3, 1997
Survey of the Incidence of Dry Cough Caused by ACE Inhibitors MOTOHIRO YAGI*, MIKIO SAITO, MANABU ABE and KATSUJI UNO Pharmacy, Suibarago Hospital õ Received October 24, 1996 Accepted September 3, 1997
Staphylococcus sp. K.pneumoniae P.mirabilis C.freundii E. cloacae Serratia sp. P. aeruginosa ml, Enterococcus avium >100ƒÊg/ml
 CHEMOTHERAPY SEPT. 1992 cefoperazone ceftazidime (CAZ), imipenem (IPM) Staphylococcus sp., Enterococcus (CPZ), faecalis, Escherichia coli, Klebsiella pneumoniae, Citrobacter freundii, Enterobacter cloacae,
CHEMOTHERAPY SEPT. 1992 cefoperazone ceftazidime (CAZ), imipenem (IPM) Staphylococcus sp., Enterococcus (CPZ), faecalis, Escherichia coli, Klebsiella pneumoniae, Citrobacter freundii, Enterobacter cloacae,



