寄稿論文 酸を用いない芳香族化合物の新しいニトロ化法 | 東京化成工業
|
|
|
- けいざぶろう さどひら
- 5 years ago
- Views:
Transcription
1 Table 1. A List of Known itrating Agents itric acid Alone or in combination with H 2 S 4, H 3 P 4, H 4, HF(BF 3 ), CF 3 C 2 H, S 3 H, CF 3 S 3 H, FS 3 H(SbF 5 ), sulfonated resins, clays, molecular sieves, graphite itrate salts Ag 3 /BF 3, K 3 /Al 3, Cu( 3 ) 2 /(C) 2, H 4 3 /(CF 3 C) 2, (H 4 ) 2 Ce( 3 ) 6 itrate esters Bu /afion-h, /BF 3, 3 Si, acetone cyanohydrin nitrate itryl compounds BF 4, PF 6, 4, (F), C 2, CF 3 C 2, PhC 2, -nitropyridinium nitrate itrogen oxides 2 3 /BF 3, 2 4 /H 2 S 4, 2 4 /Al 3, 2 5 itroalkanes C( ) 4, CH( ) 3, ( 2 ) 3 CC( ) 3 2
2 2 H KJ/mol X (Scheme 1) (Scheme 1)
3 (Scheme 2) δ δ δ 3 δ δ 3 δ 3 3 (Scheme 2) 2 (Scheme 3) H H H 2 2 H 4 H H 3 (Scheme 3) Pb( 3 ) 2 4
4 (kyodai-nitration) BF 3 Et BF 3 Et 2 Scheme 4 5
5 Table 2. The Kyodai-itration of Substituted Benzenes a - 3 CH 2 2, 0 C substrate 1 yield isomer distribution (%) o:p (o:m) = (%) ratio Et i Pr t Bu F ~ 14 0 ~ 1 86 ~ ~ ~ 53 0 ~ 3 47 ~ ~ 1.14 Br ~ 51 3 ~ 4 46 ~ ~ 1.09 C (1.1) t BuC (3.8) C 2 H (0.38) C 2 88 b (0.47) C (0.01) CEt 2 72 b (1.0) CF c (0.01) C 93 b (0.31) (0.09) HC d C (CH 2 ) CH 2 C a eactions were carried out in dichloromethane at 10 ~ 0, unless otherwise indicated. b 1,2-Dichloroethane was used as the solvent. c thanesulfonic acid was added as a catalyst. d Chloroform was used as the solvent C 3 C fum-h 3 -fum-h 2 S 4, % - 3, H (cat.), hexane, -10 >95% (Scheme 4) 7 fum-h 3 -fum-h 2 S 4, % - 3, H (cat.), CH 2 2, 0 97% 6
6 - 3 CH 2 2, 0 C (Scheme 5) H 3 -H 2 S 4, 1-/2- = 7.7 ~ 11-3, CH 2 2, 0 1-/2- = 12 ~ 16 (Scheme 5) Table 3. Comparison of the atios of the Most Abundant Isomer to the Sum of All ther Isomers Formed in the itration of aphthalenes substrate a conditions of nitration a ArH a - 3 naphthalene 10 ~ ~ ~ 70 1-methylnaphthalene ~ 13 2-methylnaphthalene ~ 4.0 a From reference 13. 7
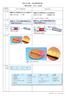
7 Figure 1. Time course of the progressive kyodai-nitration of hydrocarbon 5 to musk xylol 6. The reaction was carried out at -10 in hexane. Hydrocarbon 5 Dinitro derivatives Mononitro derivatives Musk xylol Figure 2. Dependence of the ortho-para isomer distributions on the initial concentration of chlorobenzene. Mg (Scheme 6) ( CH() 2 ) ( C() 3 ) 8
8 log [Ph /M] - 3, Mg CH 2 2, 0 C = Et t Bu ortho:meta:para yield (%) 22 : 19 : : 25 : (Scheme 6) CH(Ac) CH(Ac) 2 CH 2 2, 0 C ortho:meta:para 55 : 28 : 17 Scheme 7 (Scheme 8) C Kyodai-nitration - 3 Conventional nitration H 3 -H 2 S 4 Protection as acetal Kyodai-nitration ( - 3 ) Deprotection C C C C 2 M C 2 Η CH 2 2, 0 C M = Li K 1/2 Ca 1/2 Cu ortho:meta:para 21 : 78 : 1 26 : 73 : 1 9 : 89 : 2 30 : 68 : 2 (Scheme 7) (Scheme 8) 9
9 Scheme 9 (o : p = 3.3) (2,4- : 2,6- = 2.2 ~ 2.5) HC 13 = CH 2 2, 0 C i Pr C ortho:meta:para yield (%) 81 : 0 : : 0 : : 0 : HC (Scheme 9) (o: p = 1.2 ~ 1.5) (Scheme 10) Scheme (CH 2 ) n - 3 CH 2 2, 0 C n = 2 (CH 2 ) n ortho:meta:para yield (%) 1 69 : 4 : 27 > : 3 : 26 > : 3 : 59 >90 (Scheme 10) CH 2 C 2 2 CH 2 C 2 ortho:meta:para yield (%) 19 : 29 : : 4 :
10 C 2 Bakke 2 5 ahs 3 (Scheme 11) S 3 a - 3 ahs 3 CH 2 2, 0 C H (Scheme 11) C (Scheme 12) C (Scheme 13) 11
11 H 17-3 CH 2 2, 0 C H 3 -H 2 S H 2 3 CH 2 2, 0 C (Scheme 12) 19 H 3 S 3 H CH 2 2, 0 C 2 H 3 (S 3 H) CH 2 2, 0 C C 2 Et 3 C 23-3 M (cat.) C 2 H 4 2, 5 C - 3 M (cat.) C 2 H 4 2, 5 C 2 3 C C 2 Et (Scheme 13) C C C (Scheme 14) C (Scheme 14) CH2 2 H, 0 C 29 12
12 3 3 C C 2 2 (Scheme 15) (Scheme 15) 2 ArH 2 3 Ar H 3 2 (1) Hammett Brown σ r = ρ =
13 Figure 3. Hammett plot from the competitive kyodai-nitration. 3 (Z 0 = 2.3~2.6 V vs HE) (2) ArH ArH 3 3 (3) ArH H Ar(H) Ar (4) H H3 (5) H ArH Ar(H) Ar (6) 3 (Scheme 16) - 3 CH 2 2, 20 C CH 2 H 33 2 BF 4 69% 17% 14% no ring nitration (Scheme 16) CH 2 2, 0 C 100% (o : m : p = 8 : 24 : 68) no fragmentation 14
14 Table 4. Unusual ta rientation bserved at the Initial Stage of the KyodaiÐitration substrate Py a t b conv. c isomer proportion (%) (equiv) (min) (%) ortho : meta : para toluene : 77 : 8 chlorobenzene : 91 : 2 chlorobenzene : 71 : 20 chlorobenzene : 46 : 33 anisole : 54 : 21 a Pyridine. b eaction time. c Conversion. Scheme H H (Scheme 17) H H 3 H H Scheme Scheme 19 15
15 CH(D) 2 CH(D) 2 CD 3 34 CD(H) 3 35 H(D) CD(H) 3 36 H(D) (Scheme 18) Intramolecular 35 H /35 D 36 H /36 D kinetic isotope effect Ac X v H /v D ~6 HX v H /v D ~1.5 v H /v D = k H /k D ~6 k H /k D ~5 k H /k D =1 k H /k D =1 H CH 2 H (Scheme 19) H H CH 2 CH 2 40 CH 2 CH 2 CH CH 2 16
16 (Scheme 20) 2 2 (Scheme 20) ) Industrial Commodity Statistics Yearbook 1995, United ations, ew York, ) Mitscherich, E. Annln. Phys. Chem. 1834, 31, ) (a) Albright, L.F.; Carr..V.C.; Schmitt,.J. Eds. itration; ACS Symposium Series 623, (b) Schofield, K. Aromatic itration; Cambridge University Press: London, (c) lah, G.A.; Malhotra,.; arang, S.C. itration: thods and chanisms; VCH Publishers Inc.: ew York,
17 4) Suzuki, H.; Murashima, T.; Shimizu, K.; Tsukamoto, K. Chem. Lett. 1991, ) (a) Matsunaga, M. Chimica ggi 1994, (b) Suzuki, T.; oyori,. Chemtracts. 1997, 10, (c) Mori, T.; Suzuki, H. Synlett 1995, (d) idd, J. H. Acta Chem. Scand. 1998, 52, 11. (e) onoyama, ; Mori, T.; Suzuki, H. Zh. rg. Khim. 1998, 34, ) Suzuki, H.; Murashima, T.; Kozai, I.; Mori, T. J. Chem. Soc., Perkin Trans , ) Suzuki, H.; Murashima, T.; Shimizu, K.; Tsukamoto, K. J. Chem. Soc., Chem. Commun. 1991, ) Suzuki, H.; Mori, T. J. Chem. Soc., Perkin Trans , ) Suzuki, H.; Mori, T. J. Chem. Soc., Perkin Trans , ) (a) Bauer, A. Chem. Ber. 1891, 24, (b) Suzuki, H.; Hisatome, K.; onoyama,. Synthesis, 1999, ) Jackson, T.G.; orris, J.E.; Legendre,.C. J. rg. Chem. 1971, 36, ) Suzuki, H.; Mori, T. J. Chem. Soc., Perkin Trans , ) Eberson, L.; Hartshorn, M.P.; adner, F. Acta Chem. Scand. 1994, 48, ) Suzuki, H.; Murashima, T.; Shimizu, K.; Tsukamoto, K. Chem. Ind. (London) 1991, ) Suzuki, H.; Mori, T.; Maeda, K. J. Chem. Soc.,Chem. Commun. 1993, ) Suzuki, H.; Mori, T. J. Chem. Soc., Perkin Trans , ) Suzuki, H.; Mori, T.; Maeda, K. Synthesis 1994, ) Suzuki, H.; Yonezawa, S.; Mori, T.; Maeda, K. J. Chem. Soc., Perkin Trans , ) Suzuki, H.; Yonezawa, S.; Mori, T. Bull. Chem. Soc. Jpn. 1995, 68, ) Suzuki, H.; Tomaru, J.; Murashima, T. J. Chem. Soc., Perkin Trans , ) Suzuki, H.; onoyama,.; Tomaru, J.; Mori, T. Zh. rg. Khim., 1996, 32, ) Suzuki, H.; Tatsumi, A.; Suzuki, H.; Maeda, K. Synthesis 1995, ) Suzuki, H.; Ishibashi, T.; Marashima, T.; Tsukamoto, K. Tetrahedron Lett. 1991, 32, ) Suzuki, H.; Tatsumi, A.; Ishibashi, T.; Mori, T. J. Chem. Soc., Perkin Trans , ) Hartshorn, S..; Moodie,.B.; Shofield, K. J. Chem. Soc. B 1971, ) Suzuki, H.; Takeuchi, T.; Mori, T. J. rg. Chem. 1996, 61, ) Strazzolini, P.; Verardo, G.; Gorassini, F.; Giumanini, A.G. Bull. Chem. Soc. Jpn. 1995, 68, ) Suzuki, H.; Murashima, T.; Tatsumi, A.; Kozai, I. Chem. Lett. 1993, ) Suzuki, H.; Murashima, T. J. Chem. Soc., Perkin Trans , ) Barker, S.D.; orris,.k.; andles, D. Aust. J. Chem. 1981, 34, ) Suzuki, H.; Kozai, I.; Murashima, T. J. Chem. es. (S) 1993, ) Bakke, J.M.; Hegbom, I.; vreeide, E.; Aaby, K. Acta Chem. Scand. 1994, 48, ) Bakke, J.M.; anes, E. Synthesis 1997, ) Suzuki, H.; Iwaya, M.; Mori, T. Tetrahedron Lett. 1997, 38, ) Suzuki, H.; onoyama,. J. Chem. es. (S) 1996, ) Suzuki, H.; Suzuki, H.; to be published. 37) Suzuki, H.; onoyama,. J. Chem. Soc., Chem. Commun. 1996, ) Suzuki, H.; onoyama,. J. Chem. Soc., Perkin Trans , ) Suzuki, H.; Mori, T. J. rg. Chem. 1997, 62, ) Suzuki, H.; Takeuchi, T.; Mori, T. Bull. Chem. Soc. Jpn. 1997, 70, ) Suzuki, H.; Mori, T. Chem. Lett., 1996, ) Suzuki, H.; Mori, J. Chem. Soc., Perkin Trans , ) Suzuki, H.; Murashima, T.; Mori, T. J. Chem. Soc., Chem. Commun. 1994, ) Suzuki, H.; Mori, T. J. Chem. Soc., Perkin Trans , ) Suzuki, H Synthesis 1977, ) onoyama,.; Iwaya, M.; Suzuki, H. Tetrahedron Lett. 2000, 41, ) Tokiwa, H.; hnishi, Y. CC Crit. ev. Toxicol. 1986, 17, 23-60; World Health Statistics Annual, WH, Geneva, ) Enya, T.; Suzuki, H.; Watanabe, T.; Hirayama, T.; Hisamatsu, Y. Environ. Sci. Technol. 1997, 31, ) Kawanishi, M.; Enya, T.; Suzuki, H.; Takebe, H.; Matsui, S.; Yagi, T. Chem. es. Toxicol. 1998, 11, ) Enya, T.; Suzuki, H.; Hisamatsu, Y. Bull. Chem. Soc. Jpn. 1998, 71,
名称未設定
 1 a C CF b c 4 5 Me Me S Jacobsen's catalyst Scheme 1. eagents and conditions: (a) C (1.5 equiv), Jacobsen's catalyst (0.05 equiv), toluene, 0 C, 40 h, then trifluoroacetic anhydride (4.0 equiv), 60 C,
1 a C CF b c 4 5 Me Me S Jacobsen's catalyst Scheme 1. eagents and conditions: (a) C (1.5 equiv), Jacobsen's catalyst (0.05 equiv), toluene, 0 C, 40 h, then trifluoroacetic anhydride (4.0 equiv), 60 C,
寄稿論文 ボロンアルドール反応の新展開 | 東京化成工業
 substrate control reagent control Tf Tf amine H n-u Tf Et 3 3 n-u Tf Et syn Tf anti n Tf, Amine (1.3eq) (1.5eq) CH Cl, -78 C n n Z E CH -78 C, 1h; 0 C, 1h n H Triflate Et Tf n-u Tf c-pen Tf Tf Amine Yield
substrate control reagent control Tf Tf amine H n-u Tf Et 3 3 n-u Tf Et syn Tf anti n Tf, Amine (1.3eq) (1.5eq) CH Cl, -78 C n n Z E CH -78 C, 1h; 0 C, 1h n H Triflate Et Tf n-u Tf c-pen Tf Tf Amine Yield
 CTETS 7 8 9 11 Addition Rf Rf u Rf Rf u u : R, R 2, RS, X etc. Hydrolysisesterification Ewg Ewg H C 3 H C 2 R Ewg Ewg Ewg=C 3, C 2 R' etc. R=H or Alkyl Ewg Ewg RH Base Ewg Ewg R RH Base Ewg Ewg R R Amidation
CTETS 7 8 9 11 Addition Rf Rf u Rf Rf u u : R, R 2, RS, X etc. Hydrolysisesterification Ewg Ewg H C 3 H C 2 R Ewg Ewg Ewg=C 3, C 2 R' etc. R=H or Alkyl Ewg Ewg RH Base Ewg Ewg R RH Base Ewg Ewg R R Amidation
PowerPoint プレゼンテーション
 2004 3 3 2 3 4 5 6 7 8 9 10 T. Ito, A. Yamamoto, et al., J. Chem. Soc., Chem. Commun., 136 (1974) J. Chem. Soc., Dalton Trans., 1783 (1974) J. Chem. Soc., Dalton Trans., 1398 (1975) 11 T.Ito, A. Yamamoto,
2004 3 3 2 3 4 5 6 7 8 9 10 T. Ito, A. Yamamoto, et al., J. Chem. Soc., Chem. Commun., 136 (1974) J. Chem. Soc., Dalton Trans., 1783 (1974) J. Chem. Soc., Dalton Trans., 1398 (1975) 11 T.Ito, A. Yamamoto,
寄稿論文 オキシム誘導体を用いる新しいC-N結合生成法 | 東京化成工業
 Gabriel Beckmann sp 2 displacement Beckmann earrangement of ximes 2 S 4 2 ' ' ' Displacement on itrogen Atom of ximes Y u Y u S 2 n-bu 4 e 4 p 33% 10 mol% (n-bu) 4 e 4 5 mol% Ts 2 C 2 Cl 2, rt, 5 min e
Gabriel Beckmann sp 2 displacement Beckmann earrangement of ximes 2 S 4 2 ' ' ' Displacement on itrogen Atom of ximes Y u Y u S 2 n-bu 4 e 4 p 33% 10 mol% (n-bu) 4 e 4 5 mol% Ts 2 C 2 Cl 2, rt, 5 min e
寄稿論文 規則性無機ナノ空間が創り出す新しい触媒能 | 東京化成工業
 MCM-41 M41 MCM-41 M41 2 3 m 2 /g nm nm Mn Ti Ti H N 2 S Ti-MCM-41, H H N H H 2 2 -Urea, CH 2 Cl 2, H 2 S + S 1b 2b 3b 54%, 58% ee Ti M41 H 2 As 4 ZP 4 ZP ZS ZS 5 Me Me Me Me M41 / 15 mg MeH 1.0 mmol 89%
MCM-41 M41 MCM-41 M41 2 3 m 2 /g nm nm Mn Ti Ti H N 2 S Ti-MCM-41, H H N H H 2 2 -Urea, CH 2 Cl 2, H 2 S + S 1b 2b 3b 54%, 58% ee Ti M41 H 2 As 4 ZP 4 ZP ZS ZS 5 Me Me Me Me M41 / 15 mg MeH 1.0 mmol 89%
水素移動型不斉還元触媒|関東化学株式会社
 99% yield, 96% ee (S/C = 1000) 89% yield, 99% ee (S/C = 300) >99% yield, 97% ee () X 78% yield, 95% ee () (S,S)-u cat X = C, 3, 2 = H, CH 3, F 67-100% yield, 92-98% ee (S/C = 100-1000) 100% yield dl:meso
99% yield, 96% ee (S/C = 1000) 89% yield, 99% ee (S/C = 300) >99% yield, 97% ee () X 78% yield, 95% ee () (S,S)-u cat X = C, 3, 2 = H, CH 3, F 67-100% yield, 92-98% ee (S/C = 100-1000) 100% yield dl:meso
 4) H. Takayama et al.: J. Med. Chem., 45, 1949 (2002). 5) H. Takayama et al.: J. Am. Chem. Soc., 112, 8635 (2000). 6) H. Takayama et al.: Tetrahedron Lett., 42, 2995 (2001). 9) M. Kitajima et al: Chem.
4) H. Takayama et al.: J. Med. Chem., 45, 1949 (2002). 5) H. Takayama et al.: J. Am. Chem. Soc., 112, 8635 (2000). 6) H. Takayama et al.: Tetrahedron Lett., 42, 2995 (2001). 9) M. Kitajima et al: Chem.
03J_sources.key
 Radiation Detection & Measurement (1) (2) (3) (4)1 MeV ( ) 10 9 m 10 7 m 10 10 m < 10 18 m X 10 15 m 10 15 m ......... (isotope)...... (isotone)......... (isobar) 1 1 1 0 1 2 1 2 3 99.985% 0.015% ~0% E
Radiation Detection & Measurement (1) (2) (3) (4)1 MeV ( ) 10 9 m 10 7 m 10 10 m < 10 18 m X 10 15 m 10 15 m ......... (isotope)...... (isotone)......... (isobar) 1 1 1 0 1 2 1 2 3 99.985% 0.015% ~0% E
X線分析の進歩36 別刷
 X X X-Ray Fluorescence Analysis on Environmental Standard Reference Materials with a Dry Battery X-Ray Generator Hideshi ISHII, Hiroya MIYAUCHI, Tadashi HIOKI and Jun KAWAI Copyright The Discussion Group
X X X-Ray Fluorescence Analysis on Environmental Standard Reference Materials with a Dry Battery X-Ray Generator Hideshi ISHII, Hiroya MIYAUCHI, Tadashi HIOKI and Jun KAWAI Copyright The Discussion Group
Precisely Designed Catalysts News Letter Vol. 5 May, 2016
 recisely Designed Catalysts ews Letter Vol. 5 May, 2016 E-mail: sawamura@sci.hokudai.ac.jp Silica-SMA Silica-TI S-T Figure 1 Si 3 Si Si Si Si 2 Silica-SMA Si 3 Si Si Si Si 2 Silica-TI t Bu S S S-T t Bu
recisely Designed Catalysts ews Letter Vol. 5 May, 2016 E-mail: sawamura@sci.hokudai.ac.jp Silica-SMA Silica-TI S-T Figure 1 Si 3 Si Si Si Si 2 Silica-SMA Si 3 Si Si Si Si 2 Silica-TI t Bu S S S-T t Bu
寄稿論文 新光延試薬 | 東京化成工業
 DEAD TPP A Et C C Et Et C C Et 2 A A ex. ucleophiles (A) P 3 P 3 1 xygen itrogen Carbon Sulfur C 3 S Ts Tf Scheme 1. TPP DEAD TPP DEAD redox condensation Walden Bz Tf DEAD-TPP 87% Tf Bz Scheme 2. pk a
DEAD TPP A Et C C Et Et C C Et 2 A A ex. ucleophiles (A) P 3 P 3 1 xygen itrogen Carbon Sulfur C 3 S Ts Tf Scheme 1. TPP DEAD TPP DEAD redox condensation Walden Bz Tf DEAD-TPP 87% Tf Bz Scheme 2. pk a
寄稿論文 有機合成から現代の錬金術へ | 東京化成工業
 FAMS MT FAMS S Formaldehyde Dimethyl Dithioacetal S-xide FAMS DMS α DMS SDMS H 3 SH 3 S K 2 3 or Pyr. H 3 SH 2 l 3 Sa H 3 SH 2 S 4 Finkelstein FAMS FAMS FAMS FAMS H 2 2 H 2 + MeSH H+ H 2 1 (FAMS) 5 MT
FAMS MT FAMS S Formaldehyde Dimethyl Dithioacetal S-xide FAMS DMS α DMS SDMS H 3 SH 3 S K 2 3 or Pyr. H 3 SH 2 l 3 Sa H 3 SH 2 S 4 Finkelstein FAMS FAMS FAMS FAMS H 2 2 H 2 + MeSH H+ H 2 1 (FAMS) 5 MT
1 1 H Li Be Na M g B A l C S i N P O S F He N Cl A e K Ca S c T i V C Mn Fe Co Ni Cu Zn Ga Ge As Se B K Rb S Y Z Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb T e
 No. 1 1 1 H Li Be Na M g B A l C S i N P O S F He N Cl A e K Ca S c T i V C Mn Fe Co Ni Cu Zn Ga Ge As Se B K Rb S Y Z Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb T e I X e Cs Ba F Ra Hf Ta W Re Os I Rf Db Sg Bh
No. 1 1 1 H Li Be Na M g B A l C S i N P O S F He N Cl A e K Ca S c T i V C Mn Fe Co Ni Cu Zn Ga Ge As Se B K Rb S Y Z Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb T e I X e Cs Ba F Ra Hf Ta W Re Os I Rf Db Sg Bh
01-表紙.ai
 B 0 5 0-5 双極子核 I=1/ 2 四極子核 I 1 e Li Be B C N F Ne Na Mg 黒字はNMR 観測不可 Al Si P S Cl Ar K Ca Sc Ti V Cr MnFe Co Ni Cu ZnGaGe As Se Br Kr RbSr Y Zr NbMoTc RuRhPdAgCd In Sn SbTe I Xe Cs Ba La f Ta W Res Ir Pt
B 0 5 0-5 双極子核 I=1/ 2 四極子核 I 1 e Li Be B C N F Ne Na Mg 黒字はNMR 観測不可 Al Si P S Cl Ar K Ca Sc Ti V Cr MnFe Co Ni Cu ZnGaGe As Se Br Kr RbSr Y Zr NbMoTc RuRhPdAgCd In Sn SbTe I Xe Cs Ba La f Ta W Res Ir Pt
JAJP
 Agilent 7500ce ORS ICP-MS Glenn Woods Agilent Technologies Ltd. 5500 Lakeside, Cheadle Royal Business Park Stockport UK Agilent 7500ce ICP-MS 5 7500ce (ORS) 1 ORS 7500ce ORS ICP-MS ( ) 7500 ICP-MS (27.12
Agilent 7500ce ORS ICP-MS Glenn Woods Agilent Technologies Ltd. 5500 Lakeside, Cheadle Royal Business Park Stockport UK Agilent 7500ce ICP-MS 5 7500ce (ORS) 1 ORS 7500ce ORS ICP-MS ( ) 7500 ICP-MS (27.12
EPSON LP-S7500シリーズ 取扱説明書1 セットアップと使い方編
 A B K L N N N N A B A N B N N A B D A B A B N N N N N N N N N N N K A B E C D F G N N N A B N N A A K B C D L E L L K F A B G N C N N C D B K E A B F AC N K G A B C H K D F E B G H K I G H L J
A B K L N N N N A B A N B N N A B D A B A B N N N N N N N N N N N K A B E C D F G N N N A B N N A A K B C D L E L L K F A B G N C N N C D B K E A B F AC N K G A B C H K D F E B G H K I G H L J
42 3 u = (37) MeV/c 2 (3.4) [1] u amu m p m n [1] m H [2] m p = (4) MeV/c 2 = (13) u m n = (4) MeV/c 2 =
![42 3 u = (37) MeV/c 2 (3.4) [1] u amu m p m n [1] m H [2] m p = (4) MeV/c 2 = (13) u m n = (4) MeV/c 2 = 42 3 u = (37) MeV/c 2 (3.4) [1] u amu m p m n [1] m H [2] m p = (4) MeV/c 2 = (13) u m n = (4) MeV/c 2 =](/thumbs/73/68712188.jpg) 3 3.1 3.1.1 kg m s J = kg m 2 s 2 MeV MeV [1] 1MeV=1 6 ev = 1.62 176 462 (63) 1 13 J (3.1) [1] 1MeV/c 2 =1.782 661 731 (7) 1 3 kg (3.2) c =1 MeV (atomic mass unit) 12 C u = 1 12 M(12 C) (3.3) 41 42 3 u
3 3.1 3.1.1 kg m s J = kg m 2 s 2 MeV MeV [1] 1MeV=1 6 ev = 1.62 176 462 (63) 1 13 J (3.1) [1] 1MeV/c 2 =1.782 661 731 (7) 1 3 kg (3.2) c =1 MeV (atomic mass unit) 12 C u = 1 12 M(12 C) (3.3) 41 42 3 u
36 th IChO : - 3 ( ) , G O O D L U C K final 1
 36 th ICh - - 5 - - : - 3 ( ) - 169 - -, - - - - - - - G D L U C K final 1 1 1.01 2 e 4.00 3 Li 6.94 4 Be 9.01 5 B 10.81 6 C 12.01 7 N 14.01 8 16.00 9 F 19.00 10 Ne 20.18 11 Na 22.99 12 Mg 24.31 Periodic
36 th ICh - - 5 - - : - 3 ( ) - 169 - -, - - - - - - - G D L U C K final 1 1 1.01 2 e 4.00 3 Li 6.94 4 Be 9.01 5 B 10.81 6 C 12.01 7 N 14.01 8 16.00 9 F 19.00 10 Ne 20.18 11 Na 22.99 12 Mg 24.31 Periodic
物理予稿01-
 http://www.riken.jp/lab/dri/newpro/ 1 7 11 00 11 30 8 11 30 12 00 10 12 00 12 30 12 12 30 12 40 12 40 13 40 15 13 40 14 15 16 14 15 14 20 19 14 20 14 40 20 14 40 15 00 22 15 00 15 20 24 15 20 15 40 26
http://www.riken.jp/lab/dri/newpro/ 1 7 11 00 11 30 8 11 30 12 00 10 12 00 12 30 12 12 30 12 40 12 40 13 40 15 13 40 14 15 16 14 15 14 20 19 14 20 14 40 20 14 40 15 00 22 15 00 15 20 24 15 20 15 40 26
物理化学I-第12回(13).ppt
 I- 12-1 11 11.1 2Mg(s) + O 2 (g) 2MgO(s) [Mg 2+ O 2 ] Zn(s) + Cu 2+ (aq) Zn 2+ (aq) + Cu(s) - 2Mg(s) 2Mg 2+ (s) + 4e +) O 2 (g) + 4e 2O 2 (s) 2Mg(s) + O 2 (g) 2MgO(s) Zn(s) Zn 2+ (aq) + 2e +) Cu 2+ (aq)
I- 12-1 11 11.1 2Mg(s) + O 2 (g) 2MgO(s) [Mg 2+ O 2 ] Zn(s) + Cu 2+ (aq) Zn 2+ (aq) + Cu(s) - 2Mg(s) 2Mg 2+ (s) + 4e +) O 2 (g) + 4e 2O 2 (s) 2Mg(s) + O 2 (g) 2MgO(s) Zn(s) Zn 2+ (aq) + 2e +) Cu 2+ (aq)
Caloric Behavior of Chemical Oscillation Reactions Shuko Fujieda (Received December 16, 1996) Chemical oscillation behavior of Belousov- Zhabotinskii
 Caloric Behavior of Chemical Oscillation Reactions Shuko Fujieda (Received December 16, 1996) Chemical oscillation behavior of Belousov- Zhabotinskii (BZ) and Briggs- Rauscher (BR) reactions was followed
Caloric Behavior of Chemical Oscillation Reactions Shuko Fujieda (Received December 16, 1996) Chemical oscillation behavior of Belousov- Zhabotinskii (BZ) and Briggs- Rauscher (BR) reactions was followed
Reaction Mechanism and Liquefaction Process of Coal Yosuke MAEKAWA
 Reaction Mechanism and Liquefaction Process of Coal Yosuke MAEKAWA Fig.2 Produced gas composition of vitrinite hydrogenation at 400 Ž Fig.1 Symplified average structural model of Taiheiyo coal hydrogenation
Reaction Mechanism and Liquefaction Process of Coal Yosuke MAEKAWA Fig.2 Produced gas composition of vitrinite hydrogenation at 400 Ž Fig.1 Symplified average structural model of Taiheiyo coal hydrogenation
1) Y. Kobuke, K. Hanji, K. Horiguchi, M. Asada, Y. Nakayama, J. Furukawa, J. Am. Chem. Soc., 98, 7414(1976). 2) S. Yoshida, S. Hayano, J. Memb. Sci.,
 1) Y. Kobuke, K. Hanji, K. Horiguchi, M. Asada, Y. Nakayama, J. Furukawa, J. Am. Chem. Soc., 98, 7414(1976). 2) S. Yoshida, S. Hayano, J. Memb. Sci., 11, 157(1982). 3) J. D. Lamb, J. J. Christensen, J.
1) Y. Kobuke, K. Hanji, K. Horiguchi, M. Asada, Y. Nakayama, J. Furukawa, J. Am. Chem. Soc., 98, 7414(1976). 2) S. Yoshida, S. Hayano, J. Memb. Sci., 11, 157(1982). 3) J. D. Lamb, J. J. Christensen, J.
 The Chemistry of Shikonin, an Ancient Purple Pigment, and its Derivatives. Akira TERADA*, Yasuhiro TANOUE** and Hiroshige TANIGUCHI* The roots of Lithospermum erythrorhizon Sieb. and Zucc., (violet root,
The Chemistry of Shikonin, an Ancient Purple Pigment, and its Derivatives. Akira TERADA*, Yasuhiro TANOUE** and Hiroshige TANIGUCHI* The roots of Lithospermum erythrorhizon Sieb. and Zucc., (violet root,
untitled
 E-mail: khatano@fms.saitama-u.ac.jp Tel & Fax: 048-858-3535 Toxin Virus Bacterium Glycolipid Glycoprotein Vero toxin (Stx1 and Stx2) Side view Bottom view H H H H N H Grobotriaosyl Ceramide (Gb 3 : Galα1-4Galβ1-4Glcβ-Cer)
E-mail: khatano@fms.saitama-u.ac.jp Tel & Fax: 048-858-3535 Toxin Virus Bacterium Glycolipid Glycoprotein Vero toxin (Stx1 and Stx2) Side view Bottom view H H H H N H Grobotriaosyl Ceramide (Gb 3 : Galα1-4Galβ1-4Glcβ-Cer)
‚å™J‚å−w“LŁñ›Ä
 2007 172 FFFFFFFFF FFFFFFFFF 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 c d e cc bd b fb ag ag ed ed ed bd b b ef bf f df bd f bff d D f F d f 19 bd 20 21 F C e e f b b b 22 d d e f e f bf bd 23 24 222222222222222222222222222222222222222222222222222222222222222222222222
2007 172 FFFFFFFFF FFFFFFFFF 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 c d e cc bd b fb ag ag ed ed ed bd b b ef bf f df bd f bff d D f F d f 19 bd 20 21 F C e e f b b b 22 d d e f e f bf bd 23 24 222222222222222222222222222222222222222222222222222222222222222222222222
124dr
 ,'-bis(salicylidene)ethylenediamine Scheme 1 2 2 2 2 2 2 2 2-2 2 n 5 5 5' 3 3' 3 1 1 1 1 1 1 1 = stereogenic center 1 = bulky and/or chiral substituent Scheme 1. ,,,, Fig. 1 stepped conformation 2 A achiral
,'-bis(salicylidene)ethylenediamine Scheme 1 2 2 2 2 2 2 2 2-2 2 n 5 5 5' 3 3' 3 1 1 1 1 1 1 1 = stereogenic center 1 = bulky and/or chiral substituent Scheme 1. ,,,, Fig. 1 stepped conformation 2 A achiral
1) K. J. Laidler, "Reaction Kinetics", Vol. II, Pergamon Press, New York (1963) Chap. 1 ; P. G. Ashmore, "Catalysis and Inhibition of Chemical Reactio
 1) K. J. Laidler, "Reaction Kinetics", Vol. II, Pergamon Press, New York (1963) Chap. 1 ; P. G. Ashmore, "Catalysis and Inhibition of Chemical Reactions", Butterworths, London (1963) Chap. 7, p. 185. 2)
1) K. J. Laidler, "Reaction Kinetics", Vol. II, Pergamon Press, New York (1963) Chap. 1 ; P. G. Ashmore, "Catalysis and Inhibition of Chemical Reactions", Butterworths, London (1963) Chap. 7, p. 185. 2)
000..\..
 Bull. Nagoya Univ. Museum No. 20, 79 91, 2004 Quantitative chemical analysis of rocks with X-ray fluorescence analyzer XRF-1800 NAKAZAKI Mineko TSUBOI Motohiro KANAGAWA Kazuyo KATO Takenori SUZUKI Kazuhiro
Bull. Nagoya Univ. Museum No. 20, 79 91, 2004 Quantitative chemical analysis of rocks with X-ray fluorescence analyzer XRF-1800 NAKAZAKI Mineko TSUBOI Motohiro KANAGAWA Kazuyo KATO Takenori SUZUKI Kazuhiro
元素分析
 : このマークが付してある著作物は 第三者が有する著作物ですので 同著作物の再使用 同著作物の二次的著作物の創作等については 著作権者より直接使用許諾を得る必要があります (PET) 1 18 1 18 H 2 13 14 15 16 17 He 1 2 Li Be B C N O F Ne 3 4 5 6 7 8 9 10 Na Mg 3 4 5 6 7 8 9 10 11 12 Al Si P
: このマークが付してある著作物は 第三者が有する著作物ですので 同著作物の再使用 同著作物の二次的著作物の創作等については 著作権者より直接使用許諾を得る必要があります (PET) 1 18 1 18 H 2 13 14 15 16 17 He 1 2 Li Be B C N O F Ne 3 4 5 6 7 8 9 10 Na Mg 3 4 5 6 7 8 9 10 11 12 Al Si P
 2 1 7 - TALK ABOUT 21 μ TALK ABOUT 21 Ag As Se 2. 2. 2. Ag As Se 1 2 3 4 5 6 7 8 9 1 1 2 3 4 5 6 7 8 9 1 1 2 3 4 5 6 7 8 9 1 Sb Ga Te 2. Sb 2. Ga 2. Te 1 2 3 4 5 6 7 8 9 1 1 2 3 4 5 6 7 8 9 1 1 2 3 4
2 1 7 - TALK ABOUT 21 μ TALK ABOUT 21 Ag As Se 2. 2. 2. Ag As Se 1 2 3 4 5 6 7 8 9 1 1 2 3 4 5 6 7 8 9 1 1 2 3 4 5 6 7 8 9 1 Sb Ga Te 2. Sb 2. Ga 2. Te 1 2 3 4 5 6 7 8 9 1 1 2 3 4 5 6 7 8 9 1 1 2 3 4
untitled
 254nm UV TiO 2 20nm :Sr 5 Ta 4 O 15 3 4 KEY-1 KEY-2 (Ti,Nb,Ta) 5 KEY-1 KEY-2 6 7 NiO/ Sr 2 Ta 2 O 7 mmol h -1 g -1 20 15 10 5 H 2 O 2 H 2 O 2 0 0 2 4 6 8 10 12 NiO/Sr 2 Ta 2 O 7 The synthesis of photocatalysts
254nm UV TiO 2 20nm :Sr 5 Ta 4 O 15 3 4 KEY-1 KEY-2 (Ti,Nb,Ta) 5 KEY-1 KEY-2 6 7 NiO/ Sr 2 Ta 2 O 7 mmol h -1 g -1 20 15 10 5 H 2 O 2 H 2 O 2 0 0 2 4 6 8 10 12 NiO/Sr 2 Ta 2 O 7 The synthesis of photocatalysts
H1-H4
 42 S H He Li H He Li Be B C N O F Ne Be B C N O F Ne H He Li Be B H H e L i Na Mg Al Si S Cl Ar Na Mg Al Si S Cl Ar C N O F Ne Na Be B C N O F Ne Na K Sc T i V C r K Sc Ti V Cr M n F e C o N i Mn Fe Mg
42 S H He Li H He Li Be B C N O F Ne Be B C N O F Ne H He Li Be B H H e L i Na Mg Al Si S Cl Ar Na Mg Al Si S Cl Ar C N O F Ne Na Be B C N O F Ne Na K Sc T i V C r K Sc Ti V Cr M n F e C o N i Mn Fe Mg
Nuclear Magnetic Resonance 1 H NMR spectrum PPM
 Nuclear Magnetic Resonance 1 NMR spectrum PPM PPM X: X X: 1.5 1.5 1 1 1 1 : 1: 1 : 1.5 : 1.5 2 : 2 : 2 : 3 : 3 1 13 NMR spectrum BM PPM BM 13 NMR spectrum DEPT OSY a b c d a c 1 NMR spectrum PPM a-c a
Nuclear Magnetic Resonance 1 NMR spectrum PPM PPM X: X X: 1.5 1.5 1 1 1 1 : 1: 1 : 1.5 : 1.5 2 : 2 : 2 : 3 : 3 1 13 NMR spectrum BM PPM BM 13 NMR spectrum DEPT OSY a b c d a c 1 NMR spectrum PPM a-c a
(e ) (µ ) (τ ) ( (ν e,e ) e- (ν µ,µ ) µ- (ν τ,τ ) τ- ) ( ) ( ) ( ) (SU(2) ) (W +,Z 0,W ) * 1) [ ] [ ] [ ] ν e ν µ ν τ e µ τ, e R,µ R,τ R (2.1a
![(e ) (µ ) (τ ) ( (ν e,e ) e- (ν µ,µ ) µ- (ν τ,τ ) τ- ) ( ) ( ) ( ) (SU(2) ) (W +,Z 0,W ) * 1) [ ] [ ] [ ] ν e ν µ ν τ e µ τ, e R,µ R,τ R (2.1a (e ) (µ ) (τ ) ( (ν e,e ) e- (ν µ,µ ) µ- (ν τ,τ ) τ- ) ( ) ( ) ( ) (SU(2) ) (W +,Z 0,W ) * 1) [ ] [ ] [ ] ν e ν µ ν τ e µ τ, e R,µ R,τ R (2.1a](/thumbs/100/144882653.jpg) 1 2 2.1 (e ) (µ ) (τ ) ( (ν e,e ) e- (ν µ,µ ) µ- (ν τ,τ ) τ- ) ( ) ( ) ( ) (SU(2) ) (W +,Z 0,W ) * 1) [ ] [ ] [ ] ν e ν µ ν τ e µ τ, e R,µ R,τ R (2.1a) L ( ) ) * 2) W Z 1/2 ( - ) d u + e + ν e 1 1 0 0
1 2 2.1 (e ) (µ ) (τ ) ( (ν e,e ) e- (ν µ,µ ) µ- (ν τ,τ ) τ- ) ( ) ( ) ( ) (SU(2) ) (W +,Z 0,W ) * 1) [ ] [ ] [ ] ν e ν µ ν τ e µ τ, e R,µ R,τ R (2.1a) L ( ) ) * 2) W Z 1/2 ( - ) d u + e + ν e 1 1 0 0
Syntheses of Medium-sized Ring Amines from Bicyclic Amines Yoshio ARATA* and Miyoji HANAOKA** Conversion of bicyclic amines into medium-sized ring ami
 Syntheses of Medium-sized Ring Amines from Bicyclic Amines Yoshio ARATA* and Miyoji HANAOKA** Conversion of bicyclic amines into medium-sized ring amines (tertiary and secondary) by selective cleavage
Syntheses of Medium-sized Ring Amines from Bicyclic Amines Yoshio ARATA* and Miyoji HANAOKA** Conversion of bicyclic amines into medium-sized ring amines (tertiary and secondary) by selective cleavage
untitled
 1 / 37 5-4 6.1 1 2 / 37 1 (1) FePt AuAg (2) CdSe ZnSXY 2 O 3 X X : ZnO (3) SiO 2 (4) (5) 2 1 3 / 37 1 (1) FePt AuAg (2) CdSe ZnSXY 2 O 3 X X : ZnO (3) SiO 2 (4) (5) 3 4 / 37 1Tbits/cm 2 HD FePt FePt 110nm
1 / 37 5-4 6.1 1 2 / 37 1 (1) FePt AuAg (2) CdSe ZnSXY 2 O 3 X X : ZnO (3) SiO 2 (4) (5) 2 1 3 / 37 1 (1) FePt AuAg (2) CdSe ZnSXY 2 O 3 X X : ZnO (3) SiO 2 (4) (5) 3 4 / 37 1Tbits/cm 2 HD FePt FePt 110nm
Activation and Control of Electron-Transfer Reactions by Noncovalent Bond
 2 + 4e- + 4 + hν 2 2 1 2 20 J. Am. Chem. oc. Angew. Chem. Int. Ed. umber of Papers 15 10 5 0 1998 1999 2000 2001 2002 2003 Year : J. Am. Chem. oc. (Trost, B. M.; tanford University, UA) 3 π 1/2 k ET =
2 + 4e- + 4 + hν 2 2 1 2 20 J. Am. Chem. oc. Angew. Chem. Int. Ed. umber of Papers 15 10 5 0 1998 1999 2000 2001 2002 2003 Year : J. Am. Chem. oc. (Trost, B. M.; tanford University, UA) 3 π 1/2 k ET =
電子部品はんだ接合部の熱疲労寿命解析
 43 Evaluation for Thermal Fatigue Life of Solder Joints in Electronic Components Haruhiko Yamada, Kazuyoshi Ogawa 2 63Sn- 37Pb 95Pb-5Sn Si Cu Si 63Sn-37Pb Since automotive electronic components are used
43 Evaluation for Thermal Fatigue Life of Solder Joints in Electronic Components Haruhiko Yamada, Kazuyoshi Ogawa 2 63Sn- 37Pb 95Pb-5Sn Si Cu Si 63Sn-37Pb Since automotive electronic components are used
1/120 別表第 1(6 8 及び10 関係 ) 放射性物質の種類が明らかで かつ 一種類である場合の放射線業務従事者の呼吸する空気中の放射性物質の濃度限度等 添付 第一欄第二欄第三欄第四欄第五欄第六欄 放射性物質の種類 吸入摂取した 経口摂取した 放射線業 周辺監視 周辺監視 場合の実効線 場合
 1/120 別表第 1(6 8 及び10 関係 ) 放射性物質の種類が明らかで かつ 一種類である場合の放射線業務従事者の呼吸する空気中の放射性物質の濃度限度等 添付 第一欄第二欄第三欄第四欄第五欄第六欄 放射性物質の種類 吸入摂取した 経口摂取した 放射線業 周辺監視 周辺監視 場合の実効線 場合の実効線 務従事者 区域外の 区域外の 量係数 量係数 の呼吸す 空気中の 水中の濃 る空気中 濃度限度
1/120 別表第 1(6 8 及び10 関係 ) 放射性物質の種類が明らかで かつ 一種類である場合の放射線業務従事者の呼吸する空気中の放射性物質の濃度限度等 添付 第一欄第二欄第三欄第四欄第五欄第六欄 放射性物質の種類 吸入摂取した 経口摂取した 放射線業 周辺監視 周辺監視 場合の実効線 場合の実効線 務従事者 区域外の 区域外の 量係数 量係数 の呼吸す 空気中の 水中の濃 る空気中 濃度限度
Fig. ph Si-O-Na H O Si- Na OH Si-O-Si OH Si-O Si-OH Si-O-Si Si-O Si-O Si-OH Si-OH Si-O-Si H O 6
 NMR ESR NMR 5 Fig. ph Si-O-Na H O Si- Na OH Si-O-Si OH Si-O Si-OH Si-O-Si Si-O Si-O Si-OH Si-OH Si-O-Si H O 6 Fig. (a) Na O-B -Si Na O-B Si Fig. (b) Na O-CaO-SiO Na O-CaO-B -Si. Na O-. CaO-. Si -. Al O
NMR ESR NMR 5 Fig. ph Si-O-Na H O Si- Na OH Si-O-Si OH Si-O Si-OH Si-O-Si Si-O Si-O Si-OH Si-OH Si-O-Si H O 6 Fig. (a) Na O-B -Si Na O-B Si Fig. (b) Na O-CaO-SiO Na O-CaO-B -Si. Na O-. CaO-. Si -. Al O
RN201602_cs5_0122.indd
 ISSN 1349-1229 No.416 February 2016 2 SPECIAL TOPIC113 SPECIAL TOPIC 113 FACE Mykinso 113 SPECIAL TOPIC IUPAC 11320151231 RI RIBFRILAC 20039Zn30 Bi83 20047113 20054201283 113 1133 Bh107 20082009 113 113
ISSN 1349-1229 No.416 February 2016 2 SPECIAL TOPIC113 SPECIAL TOPIC 113 FACE Mykinso 113 SPECIAL TOPIC IUPAC 11320151231 RI RIBFRILAC 20039Zn30 Bi83 20047113 20054201283 113 1133 Bh107 20082009 113 113
RAA-05(201604)MRA対応製品ver6
 M R A 対 応 製 品 ISO/IEC 17025 ISO/IEC 17025は 試験所及び校正機関が特定の試験又は 校正を実施する能力があるものとして認定を 受けようとする場合の一般要求事項を規定した国際規格 国際相互承認 MRA Mutual Recognition Arrangement 相互承認協定 とは 試験 検査を実施する試験所 検査機関を認定する国際組織として ILAC 国際試験所認定協力機構
M R A 対 応 製 品 ISO/IEC 17025 ISO/IEC 17025は 試験所及び校正機関が特定の試験又は 校正を実施する能力があるものとして認定を 受けようとする場合の一般要求事項を規定した国際規格 国際相互承認 MRA Mutual Recognition Arrangement 相互承認協定 とは 試験 検査を実施する試験所 検査機関を認定する国際組織として ILAC 国際試験所認定協力機構
2001 Mg-Zn-Y LPSO(Long Period Stacking Order) Mg,,,. LPSO ( ), Mg, Zn,Y. Mg Zn, Y fcc( ) L1 2. LPSO Mg,., Mg L1 2, Zn,Y,, Y.,, Zn, Y Mg. Zn,Y., 926, 1
 Mg-LPSO 2566 2016 3 2001 Mg-Zn-Y LPSO(Long Period Stacking Order) Mg,,,. LPSO ( ), Mg, Zn,Y. Mg Zn, Y fcc( ) L1 2. LPSO Mg,., Mg L1 2, Zn,Y,, Y.,, Zn, Y Mg. Zn,Y., 926, 1 1,.,,., 1 C 8, 2 A 9.., Zn,Y,.
Mg-LPSO 2566 2016 3 2001 Mg-Zn-Y LPSO(Long Period Stacking Order) Mg,,,. LPSO ( ), Mg, Zn,Y. Mg Zn, Y fcc( ) L1 2. LPSO Mg,., Mg L1 2, Zn,Y,, Y.,, Zn, Y Mg. Zn,Y., 926, 1 1,.,,., 1 C 8, 2 A 9.., Zn,Y,.
untitled
 27.2.9 TOF-SIMS SIMS TOF-SIMS SIMS Mass Spectrometer ABCDE + ABC+ DE + Primary Ions: 1 12 ions/cm 2 Molecular Fragmentation Region ABCDE ABCDE 1 15 atoms/cm 2 Molecular Desorption Region Why TOF-SIMS?
27.2.9 TOF-SIMS SIMS TOF-SIMS SIMS Mass Spectrometer ABCDE + ABC+ DE + Primary Ions: 1 12 ions/cm 2 Molecular Fragmentation Region ABCDE ABCDE 1 15 atoms/cm 2 Molecular Desorption Region Why TOF-SIMS?
Juntendo Medical Journal
 The hair growth effect of minoxidil Susumu OTOMO, Ph. D. Key words : minoxidil, minoxidil sulfate, hair growth, K+-channel opener, male pattern baldness Dept. of Pharmacology, Research Center Taisho Pharmaceutical
The hair growth effect of minoxidil Susumu OTOMO, Ph. D. Key words : minoxidil, minoxidil sulfate, hair growth, K+-channel opener, male pattern baldness Dept. of Pharmacology, Research Center Taisho Pharmaceutical
 / ( ) 1 1.1 323 206 23 ( 23 529 529 323 206 ) 23 1.2 33 1.3 323 61 61 3721 3721 323 168 168 323 23 61 61 23 1403 323 111 111 168 206 323 47 111 323 47 2 23 2 2.1 34 2 2.2 2 a, b N a b N a b (mod N) mod
/ ( ) 1 1.1 323 206 23 ( 23 529 529 323 206 ) 23 1.2 33 1.3 323 61 61 3721 3721 323 168 168 323 23 61 61 23 1403 323 111 111 168 206 323 47 111 323 47 2 23 2 2.1 34 2 2.2 2 a, b N a b N a b (mod N) mod
Structural Studies of Graphite Intercalation Compounds of Fluorine by Transmission Electron Microscopy Tetsuya Isshiki, Fujio Okino, Yoshiyuki Hattori
 Structural Studies of Graphite Intercalation Compounds of Fluorine by Transmission Electron Microscopy Tetsuya Isshiki, Fujio Okino, Yoshiyuki Hattori, Shinji Kawasaki and Hidekazu Touhara Department of
Structural Studies of Graphite Intercalation Compounds of Fluorine by Transmission Electron Microscopy Tetsuya Isshiki, Fujio Okino, Yoshiyuki Hattori, Shinji Kawasaki and Hidekazu Touhara Department of
9) H. SCHMCLZRIED: Z. Elektrochem. 66 (l%1) p ) W. D. KINGERY et al.: J. Am. Chem. Soc., 42 (1959), p ) F. HUND: Z. Phys. Chem., 199 (195
 9) H. SCHMCLZRIED: Z. Elektrochem. 66 (l%1) p. 572 10) W. D. KINGERY et al.: J. Am. Chem. Soc., 42 (1959), p.394 11) F. HUND: Z. Phys. Chem., 199 (1952), p. 142 12) J. F. ELLIOTT and M. GLEISER: " Thermochemistry
9) H. SCHMCLZRIED: Z. Elektrochem. 66 (l%1) p. 572 10) W. D. KINGERY et al.: J. Am. Chem. Soc., 42 (1959), p.394 11) F. HUND: Z. Phys. Chem., 199 (1952), p. 142 12) J. F. ELLIOTT and M. GLEISER: " Thermochemistry
Report of Special Research from the National Institute for Environmental Studies, Japan NATIONAL INSTITUTE FOR ENVIRONMENTAL STUDIES
 Report of Special Research from the National Institute for Environmental Studies, Japan NATIONAL INSTITUTE FOR ENVIRONMENTAL STUDIES ) V O Cvolatile organic compounds V O C V O C 1940 10 1 1 N Ox V
Report of Special Research from the National Institute for Environmental Studies, Japan NATIONAL INSTITUTE FOR ENVIRONMENTAL STUDIES ) V O Cvolatile organic compounds V O C V O C 1940 10 1 1 N Ox V
HITACHI 液晶プロジェクター CP-EX301NJ/CP-EW301NJ 取扱説明書 -詳細版- 【技術情報編】 日本語
 A B C D E F G H I 1 3 5 7 9 11 13 15 17 19 2 4 6 8 10 12 14 16 18 K L J Y CB/PB CR/PR COMPONENT VIDEO OUT RS-232C RS-232C RS-232C Cable (cross) LAN cable (CAT-5 or greater) LAN LAN LAN LAN RS-232C BE
A B C D E F G H I 1 3 5 7 9 11 13 15 17 19 2 4 6 8 10 12 14 16 18 K L J Y CB/PB CR/PR COMPONENT VIDEO OUT RS-232C RS-232C RS-232C Cable (cross) LAN cable (CAT-5 or greater) LAN LAN LAN LAN RS-232C BE
Microsoft Word - R22.1.doc
 22.1 S 2 2 peroxide 72% M.S. Kharasch,.C. own, J. Am. Chem. Soc., 61, 2142 (1939). C 3 C 2 C 2 C 3 2 C 3 C 2 CC 3 h C 3 C 2 C 2 C 2 70% 30% C 3 C 2 C 2 C 3 2 C 3 C 2 CC h 3 C 3 C 2 C 2 C 2 98% 2%. Sing,
22.1 S 2 2 peroxide 72% M.S. Kharasch,.C. own, J. Am. Chem. Soc., 61, 2142 (1939). C 3 C 2 C 2 C 3 2 C 3 C 2 CC 3 h C 3 C 2 C 2 C 2 70% 30% C 3 C 2 C 2 C 3 2 C 3 C 2 CC h 3 C 3 C 2 C 2 C 2 98% 2%. Sing,
1) D. D. Coffman, E. E. Jenner : JACS, 76, 2685 (1954) 2) A. Weissberger et al.:" Organic Solvent", 390 (1955) 4) L. F. Fieser:" Experiments in Organi
 1) D. D. Coffman, E. E. Jenner : JACS, 76, 2685 (1954) 2) A. Weissberger et al.:" Organic Solvent", 390 (1955) 4) L. F. Fieser:" Experiments in Organic Chemi. stry ", 21 (1955) 5) W. R. Sorenson, T. W.
1) D. D. Coffman, E. E. Jenner : JACS, 76, 2685 (1954) 2) A. Weissberger et al.:" Organic Solvent", 390 (1955) 4) L. F. Fieser:" Experiments in Organic Chemi. stry ", 21 (1955) 5) W. R. Sorenson, T. W.
C el = 3 2 Nk B (2.14) c el = 3k B C el = 3 2 Nk B
 I ino@hiroshima-u.ac.jp 217 11 14 4 4.1 2 2.4 C el = 3 2 Nk B (2.14) c el = 3k B 2 3 3.15 C el = 3 2 Nk B 3.15 39 2 1925 (Wolfgang Pauli) (Pauli exclusion principle) T E = p2 2m p T N 4 Pauli Sommerfeld
I ino@hiroshima-u.ac.jp 217 11 14 4 4.1 2 2.4 C el = 3 2 Nk B (2.14) c el = 3k B 2 3 3.15 C el = 3 2 Nk B 3.15 39 2 1925 (Wolfgang Pauli) (Pauli exclusion principle) T E = p2 2m p T N 4 Pauli Sommerfeld
Fig. 3. Morse curves for the ground ( a ) and excited state ( b ) of a diatomic molecule2) Fig. 2. The various modes of energy dissipation from electr
 Color and Constitution of Organic Colorants, and Some Approach to New Chromophore Masaru MATSUOKA Fig. 3. Morse curves for the ground ( a ) and excited state ( b ) of a diatomic molecule2) Fig. 2. The
Color and Constitution of Organic Colorants, and Some Approach to New Chromophore Masaru MATSUOKA Fig. 3. Morse curves for the ground ( a ) and excited state ( b ) of a diatomic molecule2) Fig. 2. The
6 2 T γ T B (6.4) (6.1) [( d nm + 3 ] 2 nt B )a 3 + nt B da 3 = 0 (6.9) na 3 = T B V 3/2 = T B V γ 1 = const. or T B a 2 = const. (6.10) H 2 = 8π kc2
![6 2 T γ T B (6.4) (6.1) [( d nm + 3 ] 2 nt B )a 3 + nt B da 3 = 0 (6.9) na 3 = T B V 3/2 = T B V γ 1 = const. or T B a 2 = const. (6.10) H 2 = 8π kc2 6 2 T γ T B (6.4) (6.1) [( d nm + 3 ] 2 nt B )a 3 + nt B da 3 = 0 (6.9) na 3 = T B V 3/2 = T B V γ 1 = const. or T B a 2 = const. (6.10) H 2 = 8π kc2](/thumbs/92/109118076.jpg) 1 6 6.1 (??) (P = ρ rad /3) ρ rad T 4 d(ρv ) + PdV = 0 (6.1) dρ rad ρ rad + 4 da a = 0 (6.2) dt T + da a = 0 T 1 a (6.3) ( ) n ρ m = n (m + 12 ) m v2 = n (m + 32 ) T, P = nt (6.4) (6.1) d [(nm + 32 ] )a
1 6 6.1 (??) (P = ρ rad /3) ρ rad T 4 d(ρv ) + PdV = 0 (6.1) dρ rad ρ rad + 4 da a = 0 (6.2) dt T + da a = 0 T 1 a (6.3) ( ) n ρ m = n (m + 12 ) m v2 = n (m + 32 ) T, P = nt (6.4) (6.1) d [(nm + 32 ] )a
第1章 溶接法および機器
 670 ) ) ) ) 2.1 ) 42 671 Sn T m K. T m. T m. T m 2.2 JIS BAl BPd Al-Si Al-Mn KAlF -K AlF H O Ni 43 672 Sn-In Zn EU RoHS ) Pb Cd Hg JIS Z ( ) ) ) 3.1 44 673. MO HX MX H O M X (d) Young sf lf cos sl sf lf
670 ) ) ) ) 2.1 ) 42 671 Sn T m K. T m. T m. T m 2.2 JIS BAl BPd Al-Si Al-Mn KAlF -K AlF H O Ni 43 672 Sn-In Zn EU RoHS ) Pb Cd Hg JIS Z ( ) ) ) 3.1 44 673. MO HX MX H O M X (d) Young sf lf cos sl sf lf
スライド 1
 = 9.3 kcal/mol (TF) = 30.9 kcal/mol (py) G= T S = 9.3 (298 ( 41)) = 2.9 kcal/mol (TF) = 30.9 (298 ( 41)) = 18.7 kcal/mol (py) TF py Mn-Mn Toleman cone angle θ Electronic effect of PR 3 groups in LNi(CO)
= 9.3 kcal/mol (TF) = 30.9 kcal/mol (py) G= T S = 9.3 (298 ( 41)) = 2.9 kcal/mol (TF) = 30.9 (298 ( 41)) = 18.7 kcal/mol (py) TF py Mn-Mn Toleman cone angle θ Electronic effect of PR 3 groups in LNi(CO)
10-7 cm/ 50cm 10-6 cm/ 2,4-D 2 ph
 10-7 cm/ 50cm 10-6 cm/ 2,4-D 2 ph Cd 2+ Ca 2+ - Cr(OH) 4 ph ph ph ph 6 CrO 2-4 AsO 3-4 ph 9 Cd 2+ ph 6 ph 9 ph 6 9 ph B(OH) - 4 ph B(OH) 3 ph ph B(OH) - 4 ph soil organic matter ph ph ph ph 2,4-D ph 2,4-D
10-7 cm/ 50cm 10-6 cm/ 2,4-D 2 ph Cd 2+ Ca 2+ - Cr(OH) 4 ph ph ph ph 6 CrO 2-4 AsO 3-4 ph 9 Cd 2+ ph 6 ph 9 ph 6 9 ph B(OH) - 4 ph B(OH) 3 ph ph B(OH) - 4 ph soil organic matter ph ph ph ph 2,4-D ph 2,4-D
unknown heat Q known heat Q0 (a) (b) Fig. 3 Schematic drawing of output potential difference vs. time curves from a conduction-type calorimeter. Fig.
 Netsu Sokutei 13 (3) 157-164 (1986) (a) Fig. 1 (a) Assembly diagram of high temperature twinconduction-type calorimeter (overall dimensions; h=80cm; d=65cm). A, Inconel block; B, temperature homogenizing
Netsu Sokutei 13 (3) 157-164 (1986) (a) Fig. 1 (a) Assembly diagram of high temperature twinconduction-type calorimeter (overall dimensions; h=80cm; d=65cm). A, Inconel block; B, temperature homogenizing
2_R_新技術説明会(佐々木)
 % U: 6.58%, Np, Am:.5%, Pu:.% 5.8% Cs 6.5% Sr %.9%Mo 8.74% Tc.9% TODA C 8 H 7 C 8 H 7 N CH C CH N CH O C C 8 H 7 O N MIDOA C 8 H 7 DOODA NTA + HN(C 8 H 7 ) + H O DCC + SOCl + HN(C 8 H 7 ) + Cl TODA (TODA)
% U: 6.58%, Np, Am:.5%, Pu:.% 5.8% Cs 6.5% Sr %.9%Mo 8.74% Tc.9% TODA C 8 H 7 C 8 H 7 N CH C CH N CH O C C 8 H 7 O N MIDOA C 8 H 7 DOODA NTA + HN(C 8 H 7 ) + H O DCC + SOCl + HN(C 8 H 7 ) + Cl TODA (TODA)
資源と素材
 (Shigen to Sozai) Vol.116 p. 285 290 (2000) 共存硫酸塩を含む強酸性 MnSO 4 溶液中のオゾン酸化によって生成した沈殿の X 線回折および放電特性 * 西村忠久 1 梅津良昭 2 X-ray Diffraction and Discharge Behavior of Precipitates Produced by Ozone Oxidation in
(Shigen to Sozai) Vol.116 p. 285 290 (2000) 共存硫酸塩を含む強酸性 MnSO 4 溶液中のオゾン酸化によって生成した沈殿の X 線回折および放電特性 * 西村忠久 1 梅津良昭 2 X-ray Diffraction and Discharge Behavior of Precipitates Produced by Ozone Oxidation in
Antifoaming Properties of Glycols. II Yoshiro ABE, Shuichi MATSUMURA, and Yukihiro SHIBUYA Faculty of Engineering, Keio University (3-14-1, Hiyoshi, K
 Antifoaming Properties of Glycols. II Yoshiro ABE, Shuichi MATSUMURA, and Yukihiro SHIBUYA Faculty of Engineering, Keio University (3-14-1, Hiyoshi, Kohoku-ku, Yokohama) The authors studied on the antifoaming
Antifoaming Properties of Glycols. II Yoshiro ABE, Shuichi MATSUMURA, and Yukihiro SHIBUYA Faculty of Engineering, Keio University (3-14-1, Hiyoshi, Kohoku-ku, Yokohama) The authors studied on the antifoaming
% 1% SEM-EDX - X Si Ca SEM-EDX SIMS ppm % M M T 100 % 100 % Ba 1 % 91 % 9 % 9 % 1 % 87 % 13 % 13 % 1 % 64 % 36 % 36 % 1 % 34 46
 Review on Hair Analysis in the Wakayama Arsenic Case Jun KAWAI Department of Materials Science and Engineering, Kyoto University Sakyo-ku, Kyoto 606-8501, Japan Received 6 December 2014, Revised 29 December
Review on Hair Analysis in the Wakayama Arsenic Case Jun KAWAI Department of Materials Science and Engineering, Kyoto University Sakyo-ku, Kyoto 606-8501, Japan Received 6 December 2014, Revised 29 December
1) T. L. Cottrel, A. J. Matheson, Trans. Farad. Soc., 58, 2336(1962). 2) E. N. Chesnokov, V. N. Panfilov, Teor. Eksp. Khimiya, 17, 699(1981). 3) M. Ko
 1) T. L. Cottrel, A. J. Matheson, Trans. Farad. Soc., 58, 2336(1962). 2) E. N. Chesnokov, V. N. Panfilov, Teor. Eksp. Khimiya, 17, 699(1981). 3) M. Koshi, M. Yoshimura, K. Koseki, H. Matsui, "Photoacoustic
1) T. L. Cottrel, A. J. Matheson, Trans. Farad. Soc., 58, 2336(1962). 2) E. N. Chesnokov, V. N. Panfilov, Teor. Eksp. Khimiya, 17, 699(1981). 3) M. Koshi, M. Yoshimura, K. Koseki, H. Matsui, "Photoacoustic
untitled
 NPO 2006( ) 11 14 ( ) (2006/12/3) 1 50% % - - (CO+H2) ( ) 6 44 1) --- 2) ( CO H2 ) 2 3 3 90 3 3 2 3 2004 ( ) 1 1 4 1 20% 5 ( ) ( ) 2 6 MAWERA ) MAWERA ( ) ( ) 7 6MW -- 175kW 8 ( ) 900 10 2 2 2 9 -- - 10
NPO 2006( ) 11 14 ( ) (2006/12/3) 1 50% % - - (CO+H2) ( ) 6 44 1) --- 2) ( CO H2 ) 2 3 3 90 3 3 2 3 2004 ( ) 1 1 4 1 20% 5 ( ) ( ) 2 6 MAWERA ) MAWERA ( ) ( ) 7 6MW -- 175kW 8 ( ) 900 10 2 2 2 9 -- - 10
PALL NEWS vol.126 November 2017
 PALL NEWS November 2017 Vol.126 PALL NEWS vol.126 November 2017 NEW =2000 9660 41.4 MPa 24 MPa NFPA T2.06.01 R2-2001 CAT C/90/* (1x10 6 0-28 MPa 1x10 6 29 120 C 60 C 450 Pa 340 Pa 1 MPa JIS B 8356-3/ISO
PALL NEWS November 2017 Vol.126 PALL NEWS vol.126 November 2017 NEW =2000 9660 41.4 MPa 24 MPa NFPA T2.06.01 R2-2001 CAT C/90/* (1x10 6 0-28 MPa 1x10 6 29 120 C 60 C 450 Pa 340 Pa 1 MPa JIS B 8356-3/ISO
1. Precise Determination of BaAl2O4 Cell and Certification of the Formation of Iron Bearing Solid Solution. By Hiroshi UCHIKAWA and Koichi TSUKIYAMA (
 1. Precise Determination of BaAl2O4 Cell and Certification of the Formation of Iron Bearing Solid Solution. By Hiroshi UCHIKAWA and Koichi TSUKIYAMA (Central Research Laboratory, Onoda Cement Co., Ltd.,
1. Precise Determination of BaAl2O4 Cell and Certification of the Formation of Iron Bearing Solid Solution. By Hiroshi UCHIKAWA and Koichi TSUKIYAMA (Central Research Laboratory, Onoda Cement Co., Ltd.,
2 (March 13, 2010) N Λ a = i,j=1 x i ( d (a) i,j x j ), Λ h = N i,j=1 x i ( d (h) i,j x j ) B a B h B a = N i,j=1 ν i d (a) i,j, B h = x j N i,j=1 ν i
 1. A. M. Turing [18] 60 Turing A. Gierer H. Meinhardt [1] : (GM) ) a t = D a a xx µa + ρ (c a2 h + ρ 0 (0 < x < l, t > 0) h t = D h h xx νh + c ρ a 2 (0 < x < l, t > 0) a x = h x = 0 (x = 0, l) a = a(x,
1. A. M. Turing [18] 60 Turing A. Gierer H. Meinhardt [1] : (GM) ) a t = D a a xx µa + ρ (c a2 h + ρ 0 (0 < x < l, t > 0) h t = D h h xx νh + c ρ a 2 (0 < x < l, t > 0) a x = h x = 0 (x = 0, l) a = a(x,
a b b c a c Ally anion type Propargyl/allenyl anion type a b c a b c a b c a b c a b c a b c b =,, or C a b c a b c b = Gothelf, K. V.; Jørgensen, K.
 1 a b b c a c Ally anion type Propargyl/allenyl anion type a b c a b c a b c a b c a b c a b c b =,, or C a b c a b c b = Gothelf, K. V.; Jørgensen, K. A. Chem. ev. 1998, 98, 863-909 2 itrogen in the middle
1 a b b c a c Ally anion type Propargyl/allenyl anion type a b c a b c a b c a b c a b c a b c b =,, or C a b c a b c b = Gothelf, K. V.; Jørgensen, K. A. Chem. ev. 1998, 98, 863-909 2 itrogen in the middle
Surface Morphology for Poly-L-lactide Fibers Subjected to Hydrolysis Suong-Hyu Hyon Institute for Frontier Medical Sciences, Kyoto University 53, Shog
 Surface Morphology for Poly-L-lactide Fibers Subjected to Hydrolysis Suong-Hyu Hyon Institute for Frontier Medical Sciences, Kyoto University 53, Shogoin Kawaharacho, Sakyo-ku, Kyoto, 606-8507 Japan Abstract:
Surface Morphology for Poly-L-lactide Fibers Subjected to Hydrolysis Suong-Hyu Hyon Institute for Frontier Medical Sciences, Kyoto University 53, Shogoin Kawaharacho, Sakyo-ku, Kyoto, 606-8507 Japan Abstract:
Outline I. Introduction: II. Pr 2 Ir 2 O 7 Like-charge attraction III.
 Masafumi Udagawa Dept. of Physics, Gakushuin University Mar. 8, 16 @ in Gakushuin University Reference M. U., L. D. C. Jaubert, C. Castelnovo and R. Moessner, arxiv:1603.02872 Outline I. Introduction:
Masafumi Udagawa Dept. of Physics, Gakushuin University Mar. 8, 16 @ in Gakushuin University Reference M. U., L. D. C. Jaubert, C. Castelnovo and R. Moessner, arxiv:1603.02872 Outline I. Introduction:
UBE Aromatic SF5 Compounds,
 UBE Aromatic S5 Compounds, Contact: fluorine@ube.com Molecular Model of Ph UBE の S5 化合物は 多様な S5 化合物を低コストで供給できる画期的な手法で合成されています 現在 下記の S5 化合物の提供が可能です 医薬 農薬 高分子材料 エレクトロニクス材料等 お客様が求める機能材料のビルディングブロックとしてご活用ください
UBE Aromatic S5 Compounds, Contact: fluorine@ube.com Molecular Model of Ph UBE の S5 化合物は 多様な S5 化合物を低コストで供給できる画期的な手法で合成されています 現在 下記の S5 化合物の提供が可能です 医薬 農薬 高分子材料 エレクトロニクス材料等 お客様が求める機能材料のビルディングブロックとしてご活用ください
光学
 Control of Refractive Indices of Optical Polymers: Theoretical Prediction and Molecular Design Method Shinji ANDO The fundamental theory and a method to predict the refractive indices and dispersions of
Control of Refractive Indices of Optical Polymers: Theoretical Prediction and Molecular Design Method Shinji ANDO The fundamental theory and a method to predict the refractive indices and dispersions of
1. 2. (Rowthorn, 2014) / 39 1
 ,, 43 ( ) 2015 7 18 ( ) E-mail: sasaki@econ.kyoto-u.ac.jp 1 / 39 1. 2. (Rowthorn, 2014) 3. 4. 5. 6. 7. 2 / 39 1 ( 1). ( 2). = +. 1. g. r. r > g ( 3).. 3 / 39 2 50% Figure I.1. Income inequality in the
,, 43 ( ) 2015 7 18 ( ) E-mail: sasaki@econ.kyoto-u.ac.jp 1 / 39 1. 2. (Rowthorn, 2014) 3. 4. 5. 6. 7. 2 / 39 1 ( 1). ( 2). = +. 1. g. r. r > g ( 3).. 3 / 39 2 50% Figure I.1. Income inequality in the
講義ノート 物性研究 電子版 Vol.3 No.1, (2013 年 T c µ T c Kammerlingh Onnes 77K ρ 5.8µΩcm 4.2K ρ 10 4 µωcm σ 77K ρ 4.2K σ σ = ne 2 τ/m τ 77K
 2 2 T c µ T c 1 1.1 1911 Kammerlingh Onnes 77K ρ 5.8µΩcm 4.2K ρ 1 4 µωcm σ 77K ρ 4.2K σ σ = ne 2 τ/m τ 77K τ 4.2K σ 58 213 email:takada@issp.u-tokyo.ac.jp 1933 Meissner Ochsenfeld λ = 1 5 cm B = χ B =
2 2 T c µ T c 1 1.1 1911 Kammerlingh Onnes 77K ρ 5.8µΩcm 4.2K ρ 1 4 µωcm σ 77K ρ 4.2K σ σ = ne 2 τ/m τ 77K τ 4.2K σ 58 213 email:takada@issp.u-tokyo.ac.jp 1933 Meissner Ochsenfeld λ = 1 5 cm B = χ B =
学会誌カラー(目次)/目次11‐11月
 ! Σ ! ! " # $ % & ' '! ! " # !! !!! !!!!!! On the Committee for Disputes Solution on the Atomic Damages in Japan The Legal Scheme and the Guideline making about the Damages from the Accident of FukushimaAtomic
! Σ ! ! " # $ % & ' '! ! " # !! !!! !!!!!! On the Committee for Disputes Solution on the Atomic Damages in Japan The Legal Scheme and the Guideline making about the Damages from the Accident of FukushimaAtomic
無電解めっきとレーザー照射による有機樹脂板上へのCuマイクロパターン形成
 Title 無電解めっきとレーザー照射による有機樹脂板上への Cu マイクロパターン形成 Author(s) 菊地, 竜也 ; 和智, 悠太 ; 坂入, 正敏 ; 高橋, 英明 ; 飯野, 潔 ; 片山, 直樹 Citation 表面技術, 59(8): 555-561 Issue Date 2008-08 Doc URL http://hdl.handle.net/2115/36647 Type
Title 無電解めっきとレーザー照射による有機樹脂板上への Cu マイクロパターン形成 Author(s) 菊地, 竜也 ; 和智, 悠太 ; 坂入, 正敏 ; 高橋, 英明 ; 飯野, 潔 ; 片山, 直樹 Citation 表面技術, 59(8): 555-561 Issue Date 2008-08 Doc URL http://hdl.handle.net/2115/36647 Type
Donald Carl J. Choi, β ( )
 :: α β γ 200612296 20 10 17 1 3 2 α 3 2.1................................... 3 2.2................................... 4 2.3....................................... 6 2.4.......................................
:: α β γ 200612296 20 10 17 1 3 2 α 3 2.1................................... 3 2.2................................... 4 2.3....................................... 6 2.4.......................................
** Department of Materials Science and Engineering, University of California, Los Angeles, CA 90025, USA) Preparation of Magnetopulmbite Type Ferrite
 ** Department of Materials Science and Engineering, University of California, Los Angeles, CA 90025, USA) Preparation of Magnetopulmbite Type Ferrite Thin Films by Dip-Coating Method and Magnetic Properties
** Department of Materials Science and Engineering, University of California, Los Angeles, CA 90025, USA) Preparation of Magnetopulmbite Type Ferrite Thin Films by Dip-Coating Method and Magnetic Properties
CRA3689A
 AVIC-DRZ90 AVIC-DRZ80 2 3 4 5 66 7 88 9 10 10 10 11 12 13 14 15 1 1 0 OPEN ANGLE REMOTE WIDE SET UP AVIC-DRZ90 SOURCE OFF AV CONTROL MIC 2 16 17 1 2 0 0 1 AVIC-DRZ90 2 3 4 OPEN ANGLE REMOTE SOURCE OFF
AVIC-DRZ90 AVIC-DRZ80 2 3 4 5 66 7 88 9 10 10 10 11 12 13 14 15 1 1 0 OPEN ANGLE REMOTE WIDE SET UP AVIC-DRZ90 SOURCE OFF AV CONTROL MIC 2 16 17 1 2 0 0 1 AVIC-DRZ90 2 3 4 OPEN ANGLE REMOTE SOURCE OFF
03.Œk’ì
 HRS KG NG-HRS NG-KG AIC Fama 1965 Mandelbrot Blattberg Gonedes t t Kariya, et. al. Nagahara ARCH EngleGARCH Bollerslev EGARCH Nelson GARCH Heynen, et. al. r n r n =σ n w n logσ n =α +βlogσ n 1 + v n w
HRS KG NG-HRS NG-KG AIC Fama 1965 Mandelbrot Blattberg Gonedes t t Kariya, et. al. Nagahara ARCH EngleGARCH Bollerslev EGARCH Nelson GARCH Heynen, et. al. r n r n =σ n w n logσ n =α +βlogσ n 1 + v n w
i
 i ii 1 6 9 12 15 16 19 19 19 21 21 22 22 23 26 32 34 35 36 3 37 5 5 4 4 5 iii 4 55 55 59 59 7 7 8 8 9 9 10 10 1 11 iv ozein Van Marum 1801 Cruiokshank Marum 1840 Schonbein 3 / atm 1 N 1892 50 1960 1970
i ii 1 6 9 12 15 16 19 19 19 21 21 22 22 23 26 32 34 35 36 3 37 5 5 4 4 5 iii 4 55 55 59 59 7 7 8 8 9 9 10 10 1 11 iv ozein Van Marum 1801 Cruiokshank Marum 1840 Schonbein 3 / atm 1 N 1892 50 1960 1970
Microsoft Word - R15.1.doc
 15.1 5~10 C 76~86% R.A. Pacaud< C.F.. Allen, rg. Synth., Coll. Vol. 2, 336 (1943). C C 2 C 2 3 Si C 2 C 2 98% P. Boudjouk, B.-K. Kim, B.-. an, J. Chem. Educ., 74, 1223 (1997). ( ) 3 CC C 2 Ac ( ) 3 CC
15.1 5~10 C 76~86% R.A. Pacaud< C.F.. Allen, rg. Synth., Coll. Vol. 2, 336 (1943). C C 2 C 2 3 Si C 2 C 2 98% P. Boudjouk, B.-K. Kim, B.-. an, J. Chem. Educ., 74, 1223 (1997). ( ) 3 CC C 2 Ac ( ) 3 CC
(Shigen to Sozai) Vol.116 p (2000) 石炭灰フライアッシュからのゼオライトのアルカリ水熱合成と生成物の陽イオン交換特性 * 1 1 村山憲弘山川洋亮 2 3 小川和男芝田隼次 Alkali Hydrothermal Synthesis of Zeol
 石炭灰フライアッシュからのゼオライトのアルカリ水 Title 熱合成と生成物の陽イオン交換特性 Author(s) 村山, 憲弘, 山川, 洋亮, 小川, 和男, 芝田, 隼次 Citation 資源と素材 : 資源 素材学会誌, 116(4): 279-284 Issue Date 2000 URL http://hdl.handle.net/10112/5454 資源 素材学会 (http://www.jstage.jst.go.
石炭灰フライアッシュからのゼオライトのアルカリ水 Title 熱合成と生成物の陽イオン交換特性 Author(s) 村山, 憲弘, 山川, 洋亮, 小川, 和男, 芝田, 隼次 Citation 資源と素材 : 資源 素材学会誌, 116(4): 279-284 Issue Date 2000 URL http://hdl.handle.net/10112/5454 資源 素材学会 (http://www.jstage.jst.go.
Degradation Mechanism of Ethylene-propylene-diene Terpolymer by Ozone in Aqueous Solution Satoshi MIWA 1 *, 2, Takako KIKUCHI 1, 2, Yoshito OHTAKE 1 a
 Degradation Mechanism of Ethylene-propylene-diene Terpolymer by zone in Aqueous Solution Satoshi MIWA 1, 2, Takako KIKUCHI 1, 2, Yoshito TAKE 1 and Keiji TANAKA 2 ( 1 Chemicals Evaluation and Research
Degradation Mechanism of Ethylene-propylene-diene Terpolymer by zone in Aqueous Solution Satoshi MIWA 1, 2, Takako KIKUCHI 1, 2, Yoshito TAKE 1 and Keiji TANAKA 2 ( 1 Chemicals Evaluation and Research
スライド 1
 Matsuura Laboratory SiC SiC 13 2004 10 21 22 H-SiC ( C-SiC HOY Matsuura Laboratory n E C E D ( E F E T Matsuura Laboratory Matsuura Laboratory DLTS Osaka Electro-Communication University Unoped n 3C-SiC
Matsuura Laboratory SiC SiC 13 2004 10 21 22 H-SiC ( C-SiC HOY Matsuura Laboratory n E C E D ( E F E T Matsuura Laboratory Matsuura Laboratory DLTS Osaka Electro-Communication University Unoped n 3C-SiC
Time Variation of Earthquake Volume and Energy-Density with Special Reference to Tohnankai and Mikawa Earthquake Akira IKAMi and Kumizi IIDA Departmen
 Time Variation of Earthquake Volume and Energy-Density with Special Reference to Tohnankai and Mikawa Earthquake Akira IKAMi and Kumizi IIDA Department of Earth Sciences, Nagoya University (Received January
Time Variation of Earthquake Volume and Energy-Density with Special Reference to Tohnankai and Mikawa Earthquake Akira IKAMi and Kumizi IIDA Department of Earth Sciences, Nagoya University (Received January
寄稿論文 多環状エーテル系天然物の合成戦略 | 東京化成工業
 Gymnodinium breve 1 2 3 4 Gambierdiscus toxicus 4 trans 1 Nicolaou 2 1 2 Rainier CTX3C C A K B C J D E F I G K J 1 I G Na 3 S F A B C D E C B A Na 3 S D C 2 3 W X Y V Z U A' T F' E' D' C' B' S R 4 Q P
Gymnodinium breve 1 2 3 4 Gambierdiscus toxicus 4 trans 1 Nicolaou 2 1 2 Rainier CTX3C C A K B C J D E F I G K J 1 I G Na 3 S F A B C D E C B A Na 3 S D C 2 3 W X Y V Z U A' T F' E' D' C' B' S R 4 Q P
eaxys Prize lub Symposium in Japan, March 28, 2014 Profile JSPS Stephan
 16:00- - 2 2 Access to a Stable 2 2 4-Membered ing with on-kekulé nglet Biradical haracter from a Disilyne Dr. Katsuhiko Takeuchi 2007 3 2009 3 2012 3 2012 4 2012 4 (JSPS ) 2013 8 eaxys Prize lub Symposium
16:00- - 2 2 Access to a Stable 2 2 4-Membered ing with on-kekulé nglet Biradical haracter from a Disilyne Dr. Katsuhiko Takeuchi 2007 3 2009 3 2012 3 2012 4 2012 4 (JSPS ) 2013 8 eaxys Prize lub Symposium
寄稿論文 ナノスケールの孤独空間を有する包接化合物 ~カプセル分子~ | 東京化成工業
 2 Gutsche Cone 3 ebek, Jr. Cone upper rim 6a 6b Donor - AcceptorFET Fluorescence esonance Energy Transfer 4 Pd(II) Pt(II) Stang Pd(II) Pt(II) 5 10 11 12 6 12 M 12 12 12 12 Pd(II) Pt(II) 14 Dalcanale 7
2 Gutsche Cone 3 ebek, Jr. Cone upper rim 6a 6b Donor - AcceptorFET Fluorescence esonance Energy Transfer 4 Pd(II) Pt(II) Stang Pd(II) Pt(II) 5 10 11 12 6 12 M 12 12 12 12 Pd(II) Pt(II) 14 Dalcanale 7
PowerPoint プレゼンテーション
 ICP-AES, AA ppb - 1% ICP-MS ppt - 100ppm SEM-EDX 0.1% - 100% B - U XRF 0.01% - 100% Na - U EMIA ppm - 100% C, S EMGA EMIA : EMGA ppm - 100% O, N, H Carbon Sulfur Nitrigen Oxygen ISO ISO/TC 17/SC ISO 9556
ICP-AES, AA ppb - 1% ICP-MS ppt - 100ppm SEM-EDX 0.1% - 100% B - U XRF 0.01% - 100% Na - U EMIA ppm - 100% C, S EMGA EMIA : EMGA ppm - 100% O, N, H Carbon Sulfur Nitrigen Oxygen ISO ISO/TC 17/SC ISO 9556
…h…L…–…†…fi…g1
 21 1326 1328 2002 22 4375 4377 2003 34 6 848 849 2005 9 10 15 1067 1070 1996 49 133 166 2003 1 2 2 3 4 4 1 3 23 311 313 2004 5 1 118 7604 7607 1996 15 1 33 6627 6629 2000 12 649 155 167 2003 76 23 7007
21 1326 1328 2002 22 4375 4377 2003 34 6 848 849 2005 9 10 15 1067 1070 1996 49 133 166 2003 1 2 2 3 4 4 1 3 23 311 313 2004 5 1 118 7604 7607 1996 15 1 33 6627 6629 2000 12 649 155 167 2003 76 23 7007





